July 18, 2022 report
A demonstration of real-space imaging of some of the most representative forms of hydrated protons

A team of researchers with members affiliated with several institutions in China has demonstrated real-space imaging of some of the most representative forms of hydrated protons. In their paper published in the journal Science, the group describes modifying their qPlus non-intrusive atomic force microscope (AFM) to improve detection and imaging. They used it to study hydrated protons in water placed on different metal materials. Yoshiaka Sugimoto, with the University of Tokyo, has published a Perspectives piece in the same journal issue outlining the work done by the team.
As noted by Sugimoto, chemists have found it challenging to obtain desired information regarding the location and dynamics of protons during wetting—the process whereby liquids are absorbed onto solid surfaces. In this new effort, the researchers have developed a method for doing so that involves use of a modified AFM.
AFMs are microscopes that can be used to scan the surface of materials at the atomic level. A desired molecule is affixed to the tip of a probe that is then moved very close to a material under study. As the tip is moved closer, sensors monitor the forces from the material as they act on the molecule.
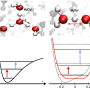
In their work, the researchers modified their microscope to study a specific material under specific conditions—in this case, a single-molecule layer of water standing on a metal surface. In their experiments, the water served as a water-wetted solid surface. Prior research has shown that under such conditions, water will behave much like ice, which means it will follow the Bernal-Fowler rules, forming a network structure with hydrogen bonds.
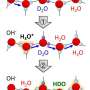
In using their modified AFM, the researchers were able to demonstrate real-space imaging of two common forms of hydrated protons; Eigen and Zundel cations. They were also able to observe D and L defects. The researchers were also able to observe coupled Eigen-Zundel interconversions involved in proton transfer, which, they note, could play a yet-to-be-understood role in electrochemical processes.
More information: Ye Tian et al, Visualizing Eigen/Zundel cations and their interconversion in monolayer water on metal surfaces, Science (2022). DOI: 10.1126/science.abo0823
Yoshiaki Sugimoto, Seeing how ice breaks the rule, Science (2022). DOI: 10.1126/science.add0841
Journal information: Science
© 2022 Science X Network