Boosting electrocatalytic ammonia synthesis of rhodium catalyst
A research team led by Prof. Zhang Haimin from the Hefei Institutes of Physical Science (HFIPS) of the Chinese Academy of Sciences has reported their discovery of dodecanethiol-modified metallic rhodium (Rh) for high-performance electrocatalytic nitrogen (N2) to ammonia.
The interface engineering approach they applied in this study, according to the team, is very helpful in developing efficient nitrogen reduction reaction (NRR) electrocatalysts for ammonia synthesis under ambient conditions. Related results were published in Nano Research.
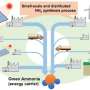
Compared with the Haber-Bosch ammonia synthesis process with demanding reaction conditions and high energy consumption, the electrocatalytic NRR can be carried out at room temperature and pressure, and the source of hydrogen is water. Therefore, it has important scientific research value and industrial application feasibility.
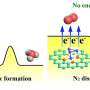
However, the non-dipole and low solubility of nitrogen make it difficult to adsorb on the catalyst surface and be activated. In addition, electrolytes are a natural proton source. Compared with N2, the protons generated by water splitting have lower activation energy, so the reaction sites are more easily occupied by protons. The number of active sites for NRR was reduced, resulting in lower ammonia yield rate.
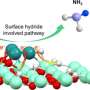
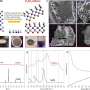
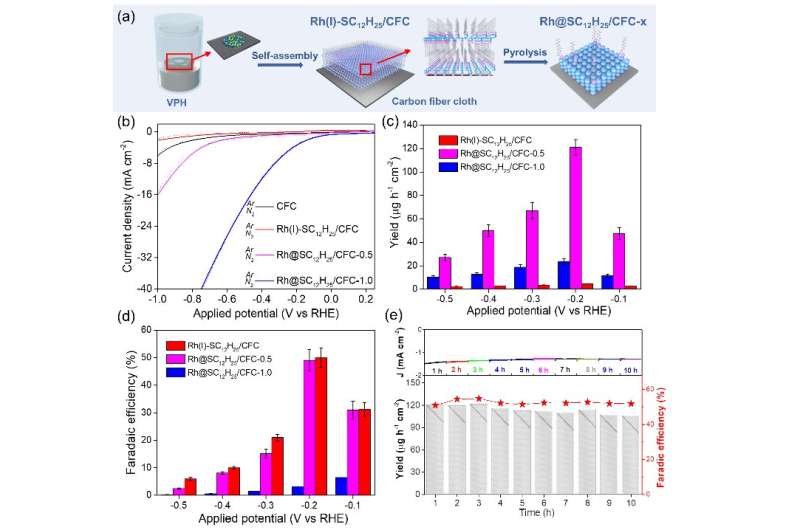
In this study, the dodecanethiol-modified Rh was fabricated via a facile saturated dodecanethiol vapor-phase hydrothermal reaction followed by low-temperature pyrolysis process. The hydrophobic dodecanethiol molecules on Rh surface produced stereo-hindrance effect, which inhibited the diffusion of water molecules or H+ to metal surface and facilitates N2 adsorption, thus improving the NRR selectivity.
Furthermore, density-functional-theory calculations unveiled that the surface hydrogen (H*) coverage and the NRR reaction energy barrier were both decreased after dodecanethiol modification, thereby greatly enhancing the NRR performance.
This study provides new insights into the effect of the metal-organic interface and H* coverage on the electrochemical NRR activity.
More information: Meng Jin et al, Interfacial engineering of metallic rhodium by thiol modification approach for ambient electrosynthesis of ammonia, Nano Research (2022). DOI: 10.1007/s12274-022-4585-y
Journal information: Nano Research
Provided by Chinese Academy of Sciences