Ultrafine ruthenium nanocrystals as an alternative to platinum for a pH-universal hydrogen evolution reaction
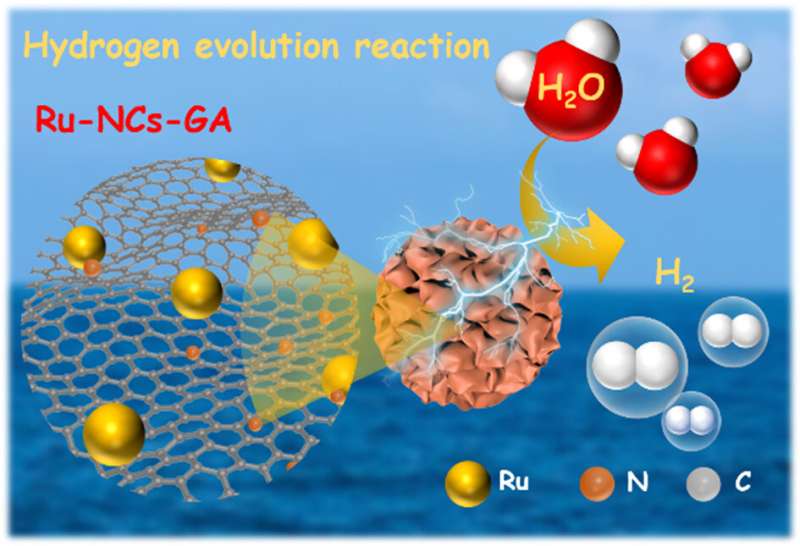
Currently, electrocatalytic water splitting is one of the most inexpensive, clean, reliable, quiet and affordable industrial-grade efficient hydrogen (H2) production technologies. However, the most effective platinum (Pt) based catalysts for the hydrogen evolution reaction (HER) are subject to high price and unsatisfactory stability. Therefore, low cost, high efficiency and high stability HER electrocatalysts are urgently needed to replace Pt based electrocatalysts.
Taking full account of the promising activity, robustness and reasonable price of ruthenium and the high surface area and excellent conductivity of graphene-based advanced carriers, Ru-carbon nanocomposites can be a feasible option for HER. However, it is still a challenge to further improve the catalytic performance of Ru-carbon nanocomposites.
Recently, a research team led by Prof. Yu Chen from Shaanxi Normal University, China designed a high-quality N-doped graphene aerogel supporting Ru nanocrystal (Ru-NCs/N-GA) nanocomposites. The reaction precursors of graphene oxide (GO) and ruthenium(III)-polyallylamine (RuIII-PAA) complex uniformly anchored in a graphene aerogel serve as the N source and Ru source.
The uniform adsorption of the RuIII-PAA complex can led to the homogeneous dispersion and ultrafine size of Ru nanocrystals on the graphene aerogel under high-temperature processing. Ru-NCs/N-GA with a porous structure and low Ru content (10 w.t.%) exhibits a comparable HER activity with 20 w.t.% Pt/C in both alkaline and acidic mediums.
Overall, economically feasible Ru-NCs/N-GA nanocomposites with 10 wt.% Ru are highly promising substitutes for commercial Pt/C electrocatalyst for HER. The results were published in Chinese Journal of Catalysis.
More information: Yu Ding et al, Nitrogen-doped graphene aerogel-supported ruthenium nanocrystals for pH-universal hydrogen evolution reaction, Chinese Journal of Catalysis (2022). DOI: 10.1016/S1872-2067(21)63977-3
Provided by Chinese Academy of Sciences




















