Intersecting light beams key in transformative 3D printer potential
QUT researchers have used intersecting light beams to control chemical reactions in an advanced material, paving the way for future use in 3D printers that print entire layers at a time instead of single points.
QUT's Centre for Materials Science interdisciplinary research team, made up of Dr. Sarah Walden, Leona Rodrigues, Dr. Jessica Alves, Associate Professor James Blinco, Dr. Vinh Truong, and ARC Laureate Fellow Professor Christopher Barner-Kowollik, have published their research in Nature Communications.
Dr. Walden said light was a particularly desirable tool for activating chemical processes, because of the precision it offered in starting a reaction.
"Most of the work QUT's Soft Matter Materials Group researchers have done in the past with light has been to use a laser beam to start and stop a chemical reaction along the entire volume where the light strikes the material," Dr. Walden said.
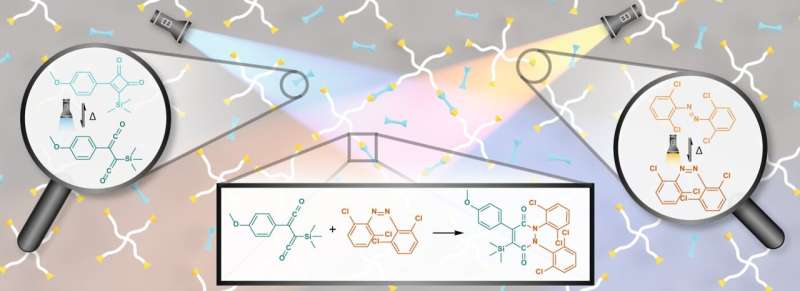
"In this case, we have two different colored light beams, and the reaction only occurs where the two beams intersect. We use one color of light to activate one molecule, and the second color of light to activate another molecule. And where the two light beams meet, the two activated molecules react to form a solid material.
"Normally, in a 3D printer, the inkjet moves around in two dimensions, slowly printing one 2D layer before moving up to print another layer on top. But using this technology, you could have a whole two-dimensional sheet activated, and print the entire sheet at once."
Professor Barner-Kowollik said such two color activated materials are currently very rare. "This project is about proving the viability of the ink for future generation of printers," he said.
Professor Barner-Kowollik, whose career is focused on the power and possibilities of light in materials science, was recently recognized with Australia's highest prize for chemistry, the 2022 David Craig medal, awarded by the Australian Academy of Science.
Professor Barner-Kowollik said one of the challenges of the project was to find two molecules that could be activated by two different colors of light and then have them react together.
"This is where the innovation comes from," Professor Barner-Kowollik said. "You want a molecule to be activated with one color of light but not the other color, and vice versa. That's not easy to find, it's actually quite hard to find."
Dr. Truong, after much work, was able to find two molecules that reacted to the lights in the required manner and combined to form a very solid material.
"In our chemical design, both light activated processes are reversible," Dr. Truong said. "Hence we can control exactly when and where the solid material may form."
More information: Sarah L. Walden et al, Two-colour light activated covalent bond formation, Nature Communications (2022). DOI: 10.1038/s41467-022-30002-6
Journal information: Nature Communications
Provided by Queensland University of Technology



















