How globalization could be making human parasites more virulent
Parasites that cause severe diarrhea are likely to become more virulent because of the speed at which they are exchanging their DNA and evolving—according to new research from the University of East Anglia.
Researchers studied the genomes of Cryptosporidium parvum—a zoonotic parasite that causes severe diarrhea in both humans and animals.
They found that different lineages of these parasites are increasingly exchanging their DNA, which has helped the parasite evolve faster—potentially resulting in more virulent and better-adapted strains.
The new study shows that the Cryptosporidium parvum lineages have exchanged more DNA in the last 200 years than in all the time before.
And they put this down to globalization and our increasingly close contact with animals which increases the rate of spill-over events.
One of the lead researchers, Prof. Cock Van Oosterhout, from UEA's School of Environmental Sciences, said: "Cryptosporidium is an important genus of zoonotic parasites, and it is one of several microorganisms that cause diarrheal disease in both humans and some livestock. In humans, it is responsible for around 57,000 deaths each year, 80% of which are among children under five years old.
"The vast majority of these are in low-income countries, but outbreaks also occur in the U.K. and elsewhere in Europe. There is no effective drug or vaccine available, so understanding the transmission and evolution of this parasite is critical.
"We know that there are several lineages of the Cryptosporidium parvum parasite, but we wanted to understand more about how they evolve, and particularly, why this species may be becoming more virulent than it was in the past."
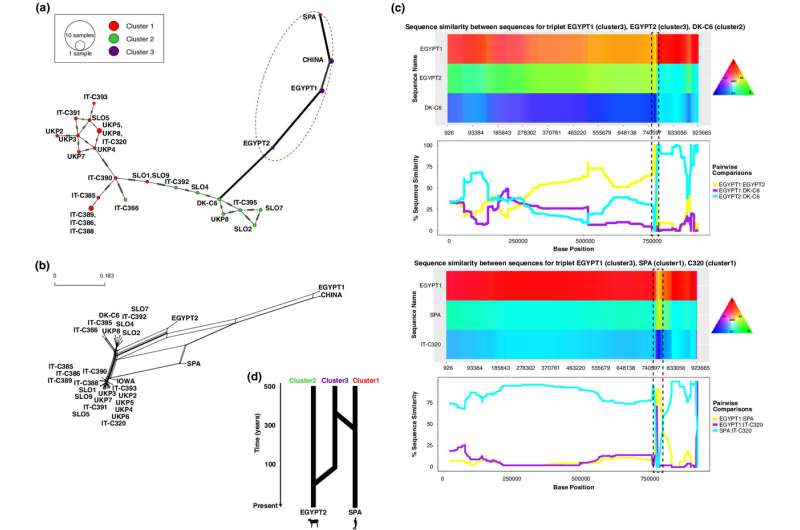
The team, led by UEA in collaboration with scientists in Italy and Australia, used whole genome sequence analysis to find out more about these genetic exchanges.
They compared 32 whole genome sequences from human and animal strains collected across Europe, U.S., Egypt, and China.
Prof. Van Oosterhout said: "We found that different lineages of these parasites are increasingly exchanging their DNA. In the last 200 years, around 22 percent of the genome of these parasites has been exchanged.
"This is significantly more than the DNA they have exchanged in all the time before. The genes that are involved in virulence seem to be particularly affected by such genetic exchanges. The genome of parasites that are infecting humans possess some DNA of parasites normally found in cows and lamb.
"We believe that these genetic exchanges help the parasite to evolve faster, and that this can result in more virulent and better-adapted human parasites. This is really important because, as COVID-19 has shown us, human parasites can evolve rapidly.
"We hypothesize that the rate of evolution is further accelerated because the parasite can evolve in multiple host species. This means that novel adaptations that have evolved in parasites that infect one host species can now be used by parasites that infect another species of hosts.
"The increased connectivity in our globalized world and close contact between humans and domesticated animals increases the rate of spill-over and spillback events.
"Using whole genome sequence analysis, we can now study these genetic exchanges and identify when and where they are taking place. This will help us better control these emerging infectious diseases of zoonotic parasites and pathogens," he added.
"Recent genetic exchanges and admixture shape the genome and population structure of a zoonotic parasite" is published in the journal Molecular Ecology.
More information: Giulia I. Corsi et al, Recent genetic exchanges and admixture shape the genome and population structure of the zoonotic pathogen Cryptosporidium parvum, Molecular Ecology (2022). DOI: 10.1111/mec.16556
Journal information: Molecular Ecology
Provided by University of East Anglia