Uniform single atomic sites anchored in graphdiyne for benzene hydroxylation to phenol
For single-atom catalysts (SACs), the catalyst supports are not only anchors for single atoms, but also modulators for geometric and electronic structures, which has an important impact on the catalytic performance. Selecting an appropriate support to prepare SACs with uniform coordination environments is critical for achieving optimal performance and clarifying the relationship between the structure and the property of SACs.
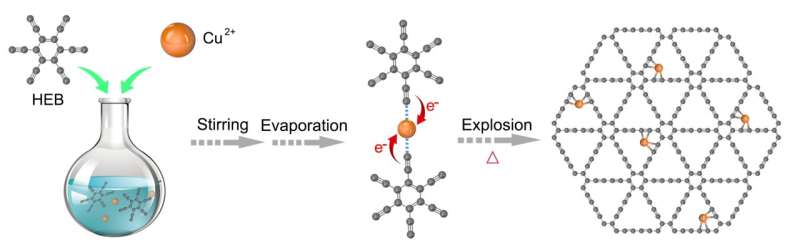
Graphdiyne (GDY), a new two-dimensional periodic carbon allotrope with an atom-thick layer, which was first synthesized by Prof. Yuliang Li in ICCAS, China, is composed of sp-hybridized carbon atoms in diacetylenic and sp2-hybridized carbon atoms in benzene rings. The unique alkyne-rich structure of GDY makes it an ideal support for anchoring single atoms due to the uniformly distributed pores and large binding energies to metal atoms via the strong d-π interaction. Taking advantage of the above characterizations of GDY, Dr. Changyan Cao and Dr. Feng He from ICCAS present an efficient and simple strategy for fabricating Cu single atoms anchored on GDY (Cu1/GDY) with uniform Cu1-(sp)C4 single sites under mild conditions.
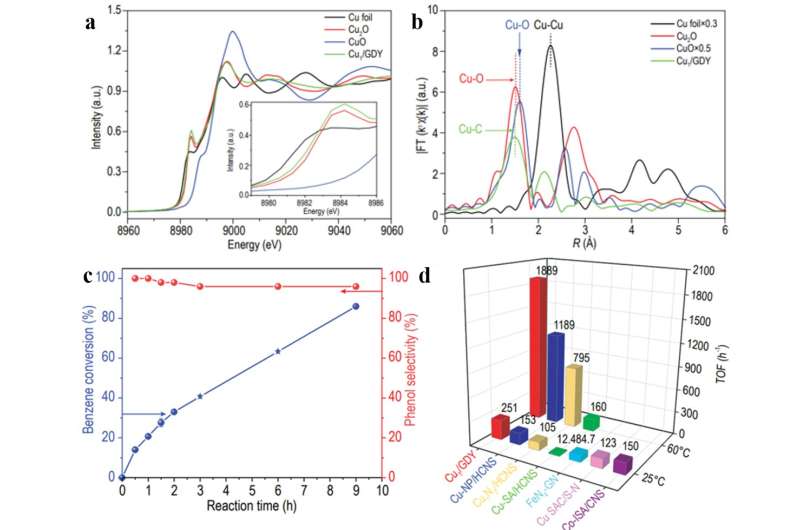
By using synchrotron radiation X-ray absorption spectroscopy, X-ray photoelectron spectroscopy, and density functional theory (DFT) calculation, it is proved that Cuδ + (0 < δ < 1) atoms are anchored on GDY in Cu1-(sp)C4 coordination environment. Cu1/GDY demonstrated excellent catalytic performance for benzene oxidation to phenol using H2O2. The calculated turnover frequency (TOF) is approximately 251 h− 1 at room temperature and 1889 h− 1 at 60 °C, which is significantly higher than previously reported catalysts under the same reaction conditions.
Furthermore, even with a high benzene conversion of 86% , high phenol selectivity (96%) is maintained, which can be ascribed to the hydrophobic and oleophilic surface nature of Cu1/GDY for benzene adsorption and phenol desorption. Synchrotron X-ray absorption spectroscopy, Fourier transform infrared absorption spectroscopy and density functional theory show that the Cu1-C4 active site can more effectively activate H2O2 to form Cu=O bond, which is an important active intermediate for the oxidation of benzene to phenol. The intrinsic higher activity of Cu1/GDY compared with other Cu SACs with nitrogen coordination structures is clarified by DFT calculations of Cu-3d band center.
This work not only presents an efficient route for fabricating GDY-supported metal SACs with uniform metal-C4 centers, but also provides a promising benzene hydroxylation catalyst for phenol production with H2O2.
The research was published in National Science Review.
More information: Jia Yu et al, Uniform Single Atomic Cu1-C4 Sites Anchored in Graphdiyne for Hydroxylation of Benzene to Phenol, National Science Review (2022). DOI: 10.1093/nsr/nwac018
Provided by Science China Press