DNA nanostructures for targeting cellular surface receptors
A broad subset of parameters, which include cell signaling modulation and receptor binding efficiency, can be explored using programmable DNA nanostructures. Glenn Cremers shows how these structures can uncover important cell signaling mechanisms, provide insights for the design of synthetically engineered communication networks and facilitate the synthesis of precision medicines.
In the human body approximately 200 specialized cells are formed starting from only one cell, the fertilized egg. To achieve this level of complexity and maintain correct cellular organization, cells extensively rely on signaling pathways. These interconnected signal-transduction pathways evolved around intricate receiver modules, receptors, that have the ability to receive and interpret external stimuli and ultimately translate them to an output.
Multivalent interaction
Receptors are located both intracellular as well as on the surface of cells and are activated by binding of their cognate ligand: the sender. However, the simplicity of the basic modules (i.e. receptors and ligands) that regulate cellular signaling does not directly translate to the extremely complex cellular signaling pathways they are associated with.
To control the complex signaling pathways, cells are heavily dependent on spatio-temporal dynamics of ligands and activated effectors. More specifically, cellular communication does not solely rely on the binding of a single ligand to a single receptor but requires the simultaneous binding of multiple receptors to their endogenous ligands, resulting in the formation of a multivalent interaction.
Actively regulating the number of ligands involved in cellular signaling increases the sensitivity of ligand recognition and, in addition, results in selective recognition of different cell-types with increased functional affinity.
Therapeutically relevant receptors
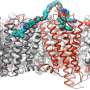
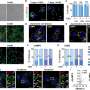
In his thesis, Cremers investigated the role of multivalency in selective recognition of cells based on a receptor-density threshold by employing the DNA origami method. Specifically, he relied on a 150 nm DNA nanorod site-specifically functionalized with a specific number of antibodies to achieve a multivalent interaction.
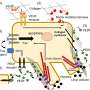
In addition, Cremers explored the determinants that modulate cellular accessibility of these DNA nanostructures and systematically studied tunable design parameters of antibody-functionalized DNA nanostructures binding to therapeutically relevant receptors.
Glenn Cremers defended his Ph.D. thesis, titled "Targeting cellular surface receptors using DNA nanostructures" on April 11th.
Provided by Eindhoven University of Technology