Mining for GEMs: Nature's precious tool to treat diseases
Inherent within all humans lies the ability to treat many of the most common diseases. Hidden compounds—or natural products—wait to be discovered, decoded, and restored for this purpose.
Chemists like Jason Crawford, associate professor of chemistry and associate professor of microbial pathogenesis, have been studying these natural products for years. Known as specialized metabolites, these small molecules are derived from our metabolism and are widely used as drugs or drug leads to treat a variety of diseases. Evolutionarily crafted into perfect structures over millions of years, they regulate a wide range of biological phenomena. The problem is most of these molecules are not available to us because they remain unknown.
In his lab at the Yale Institute of Biomolecular Design and Discovery on West Campus, Crawford and his research group of graduate students and postdocs hunt for genetically encoded molecules, or GEMs, within a sea of molecules within the human (or host) body. GEMs are important because they can be repurposed into drugs to treat cancers, infectious diseases, inflammatory bowel disease (IBD), psoriasis, and many autoimmune diseases.
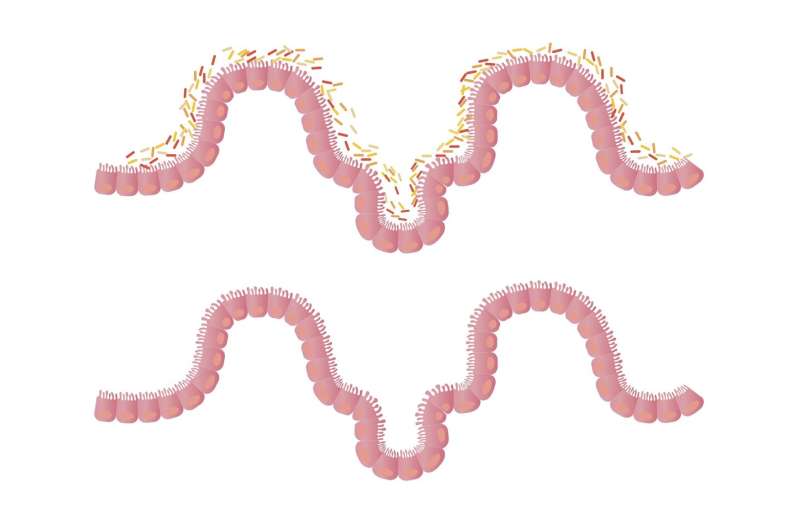
The Crawford Lab studies metabolism at the host-microbe interface. Most of the microbes they look at are either known human bacterial pathogens or environmental isolates from IBD patients collected by his collaborators in the Yale Immunobiology department. This collection of disease-associated bacteria often produces unique GEMs that positively or negatively modulate disease severity.
We recently spoke with Crawford about his research on natural molecules and their potential medicinal properties. This interview has been edited for clarity and space.
Can you share one of your latest discoveries?
What I'm most excited about right now is our recent findings of a new human and mouse metabolic pathway in this cell type called the macrophage. It's an essential innate immune cell type. Prior to our decoding efforts, mutations in that gene were linked to a wide variety of inflammatory diseases, like arthritis, IBD, and leprosy, a type of microbial infection. We also found that the gene facilitates the clearance of microbial infection, meaning if you have a dysfunctional copy, you're much more susceptible to infection.
When we decoded the pathway, we found you could add the product of the pathway—the compound—back, treat animals, and largely restore wild-type activity. What's nice about that is where gene editing is still largely experimental and not yet available to human patients with genetic defects, molecules, however, are available to treat a wide variety of inflammatory diseases.
You and a team of Yale researchers recently received an $8.5M grant from the National Institute of General Medical Sciences of the National Institutes of Health to explore the role of microbial metabolites in the human gut. Congratulations! Tell us more about this initiative.
In this project, we're trying to identify small molecules that regulate 'orphan' G-protein-coupled receptors (GPCRs). GPCRs are the most drugged receptor family in the pharmaceutical industry.
We call a subset of those 'orphan,' meaning we don't know what molecules control those receptors. These receptors often determine new biological phenomena. The molecules that manage them can tell us about the chemical signaling processes that we do not know yet. This grant particularly focuses on orphan GPCRs and our efforts to decode the molecules that regulate them.
The goal is, let's say, five years later, we have identified three different sets of molecules that regulate three different orphan GPCRs. That could open up three completely new areas of chemical signaling in humans. Those might also be druggable targets based on the history of drugging GPCRs for the biological processes.
So the grant itself is an academic endeavor to decode the unknown. The pharmaceutical relevance downstream would be to drug these new biological processes to treat diseases.
Can you speak about how microbiota—the bacteria in our gut—interacts with our immune system and your work in that space?
We're investigating compounds as therapeutics for inflammatory disease in collaboration with Richard Flavell (Immunobiology). We have at least five collaborations with Richard's lab in metabolite analysis at the molecular level to identify how microbiome members regulate unexplored areas of inflammatory signaling.
What's important to know about these efforts is that microbes use a diverse array of molecular mechanisms to regulate inflammatory processes, and we only know about some of them. So, if we screen the microbiome for small molecules that regulate noncanonical signaling outcomes, then we can discover the responsible molecules and repurpose them as 'molecular probes' to try to tease apart the signaling pathway. These efforts could allow us to discover new areas of inflammation. Once we discover them, dysregulation of those same processes could underlie different inflammatory diseases. We must understand them first before we can figure out how to treat them. Connecting microbes to these processes tells us which microbiome members either exacerbate or ameliorate the associated inflammatory diseases.
One of the inflammatory programs that we're studying is extra-intestinal inflammatory immunopathologies, which is associated with Crohn's disease. Crohn's disease is one of the inflammatory bowel diseases, but a subset of those patients has extra-intestinal inflammatory conditions, and the microbiome also mediates those.
We question how the microbiome can regulate inflammation far away from the gut. One way is to produce molecules, and then those molecules access what we call 'systemic circulation." The molecules then can regulate inflammation distally from where they were produced. There are several areas the lab is very heavily focused on. One is inflammation, and many of these projects are collaborative.
What other ways is the Crawford lab focused on inflammation?
We still have a study with Noah Palm (Immunobiology) and Seth Herzon (Chemistry), where we've decoded additional genotoxic pathways from the gut microbiome.
With our previous pathways work, Seth and I decoded a pathway associated with colorectal cancer initiation. We've recently decoded additional pathways from microbiome members that are causing tumor formation. Some might think our earlier work on colibactin was a one-off, but we've now decoded multiple pathways that are associated with cancer. Noah discovered bacteria that regulate tumor formation in mice, whereas our contribution was figuring out what molecules are responsible for regulation.
So how does this circle back to inflammation? The genotoxicity of microbes causes cellular damage, and cellular damage is another way of regulating inflammation.
What's the bigger picture? You say you've decoded some pathways that cause tumor formation. What's next?
Well, I see it like smoking, except the people with genotoxic microbes are involuntarily 'smoking' in their gut. So we have a $1.3M grant from the Department of Defense to develop what we call "Colirette." It's like Nicorette in the sense that we're developing probiotics you can take to selectively eliminate genotoxic bacteria from the gut.
Over the next three years, we will develop these probiotics and do the preclinical work needed to show proof of concept for further commercial development.
If you had unlimited resources, what further research would you pursue?
This is the question we always dream about, right, where somebody comes up and gives you a pile of money, and you don't have to write grants anymore.
I'm still very excited about the inflammation area, and our approaches, I think, are the right approaches. I would systematically expand them if I had unlimited resources because even human metabolism remains a surprisingly undefined area.
If these pathways are associated with the regulation of inflammation, then dysregulation of those pathways naturally would be associated with inflammatory disease. There are many inflammatory diseases, and we do not do a great job of treating them. We typically treat the symptoms.
But if you figure out these new pathways, you can add the molecules back and restore some level of homeostasis, and I love that these are drugs, except they're not the traditional drugs that pharma makes. They're natural. They belong within us already, and we're restoring a natural process.
We can add back the natural compound instead of an artificial one, which often has off-target effects. Because I'm more in the natural product field, I like the idea of using natural molecules to treat disease.
Provided by Yale University