Lighting up chemical reactions for sustainable drug production
Researchers have discovered a new approach for using visible-light to structurally modify bioactive molecules in chemical reactions, providing a possible sustainable alternative to traditional methods that could be used in the design and production of new medicines.
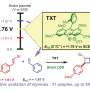
Scientists from the University of Nottingham's School of Chemistry, based at the GSK Carbon Neutral Laboratory are exploring the development of new sustainable ways to make chemicals by exploiting the green and inexhaustible energy of visible-light, as an alternative to traditional chemical methodologies requiring the use of toxic reagents and heavy metals. Their latest research "Visible Light-driven conjunctive olefination' has been published today in Nature Chemistry.
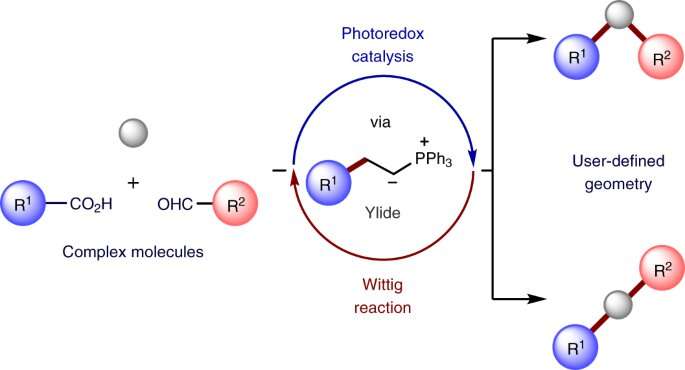
The invention of novel drugs and agrochemicals often involves investigation of several structural analogs to refine their bioactivity or other key chemical properties. Chemists often access these analogs through chemical modification of complex bioactive molecules, a process defined as "late-stage functionalisation." However, only a fraction of the possible analogs is accessible through traditional chemical methods, which often use toxic or expensive reagents and catalysts. Many catalysts rely on using heavy metals which are well-known environmental pollutants due to their toxicity, persistence in the environment, and bioaccumulative nature.
Late-stage functionalisation is an important tool for drug design. This research offers a new approach that is very modular, practical and sustainable. This new approach will significantly expand the structures available to scientists and has the potential for extensive application in the design and production of new drugs.
Dr. Mattia Silvi led the research and said: "We have now discovered a fundamentally new approach to structurally modify bioactive molecules handling them as practically as if they were Lego building blocks. Our approach exploits the sustainable energy of visible light, avoids the use of heavy metals and has general applicability, thereby opening access to exceptional room for the design of new derivatives. The results of our study could significantly impact the way scientists design new analogs of complex bioactive molecules, providing a new a new powerful tool for the development of the medicines of tomorrow."
More information: Dario Filippini et al, Visible light-driven conjunctive olefination, Nature Chemistry (2021). DOI: 10.1038/s41557-021-00807-x
Journal information: Nature Chemistry
Provided by University of Nottingham