August 11, 2021 report
Newly-synthesized AM-III carbon is hardest and strongest amorphous material to date
A team of researchers affiliated with a host of institutions across the globe has synthesized an AM-III carbon that is the hardest and strongest amorphous material created to date. In their paper published in the journal National Science Review, the group describes the process they used to create their new material and suggest possible uses for it.
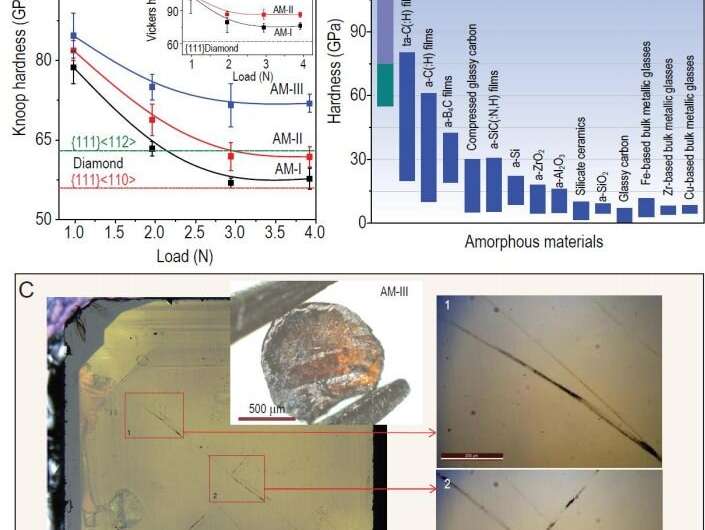
In this new effort, the researchers set out to create a new kind of glass that would be exceptionally strong. To that end, they subjected fullerenes to very high temperatures and enormous pressures and, in so doing, produced what they have called AM-III—a type of glass with crystals in it that measures higher on the Vickers hardness test than many diamonds.
When looking at a diamond under a microscope, the carbon atoms and molecules that make up its crystalline structure are lined up very neatly—glass on the other hand has very little order. This difference explains why diamonds are so hard and why glass is so easily shattered. Prior research has shown that diamonds can be made by exposing graphite to high temperatures and pressure—similar to the way they are created by nature. In this new work, the researchers instead used fullerenes—structures made of carbon in the form of hollow cages. They also slowed down the process, heating and squeezing their material for approximately 12 hours, a move to prevent the material from forming into diamond.
The resulting material, AM-III carbon, is yellowish, with no defined structure, and is very strong—it scored 113 gigapascals on the Vickers hardness test, higher than some diamonds, which average just 100 gigapascals. The researchers note that AM-III is approximately ten times as hard as steel and should be quite a bit better at stopping bullets than most vest technology. To prove its toughness, they used one sample to cut a deep scratch into a diamond. The researchers note that the toughness comes about from the material's makeup—it has micro-structures that are orderly like crystals, along with unordered glass, which makes it part glass and part crystal. It also makes the material a semiconductor with a bandgap range similar to silicon. Because of that, the researchers suggest their new material could prove useful in solar panel products.
More information: Shuangshuang Zhang et al, Discovery of carbon-based strongest and hardest amorphous material, National Science Review (2021). DOI: 10.1093/nsr/nwab140
© 2021 Science X Network