Modulation to sonoluminescence achieved in presence of carbon nano-dots in water
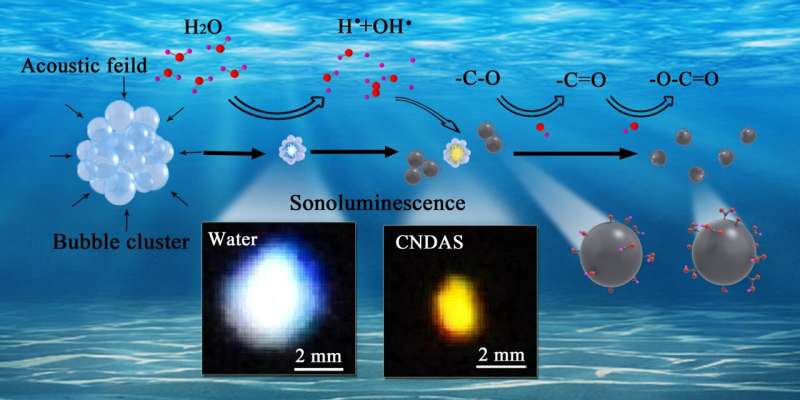
Researchers led by Prof. Xu Wen from the Hefei Institutes of Physical Science (HFIPS) of the Chinese Academy of Sciences, along with their collaborators from Chongqing Medical University, recently investigated the influence of carbon nanodots (CNDs) on sonoluminescence effect, and they found that the color of sonoluminescence driven by intense ultrasound could be altered from ultraviolet-blue in water into orange by the presence of CNDs in water.
"It shows bright orange color," said Prof. Xu, who led the team, "and it can even be seen by naked eyes."
Under the action of the focused intense ultrasound, if the negative acoustic pressure is larger than the cavitation threshold of a liquid, the cavitation takes place and the bubbles can be induced in the liquid. These bubbles undergo slow expansion and rapid collapse. At the moment of bubble collapse, light flashes can be generated and observed, which is known as the sonoluminescence. In addition, hydroxyl radicals play an important role in the sonoluminescence effect in water.
In this work, the modulation was so strong that the color of sonoluminescence could be altered significantly. The researchers measured the sonoluminescence spectra and pulses in water as well as in CND aqueous solution, and examined the changes taken place to CNDs after sonoluminescence experiment.
"Thanks to the coupling of the C-based bonds on CNDs with free hydroxyl radicals generated during the cavitation and sonoluminescence processes, we had the modulation," said Xu, "these findings provide experimental evidence for the understanding of the mechanism of the sonoluminescence effect."
On the other hand, ultrasonic treatment is a common technique applied for the synthesis of carbon nanomaterials. This time, the researchers found that the intense ultrasound brought both physical and chemical effects on CNDs.
Physically, intense ultrasound could reduce the size and result in a better crystallization of the carbon core along with a more uniformed dispersion of CNDs. Chemically, hydroxyl radical generated by the cavitation and sonoluminescence could lead to chain-like oxidation reactions with the functional groups on CNDs to form more carboxyl groups. These results are helpful for designing, synthesizing and modulating the fluorescent carbon dots.
More information: Dan Song et al, Influence of carbon nano-dots in water on sonoluminescence, Nanoscale (2021). DOI: 10.1039/D1NR02194J
Journal information: Nanoscale
Provided by Chinese Academy of Sciences