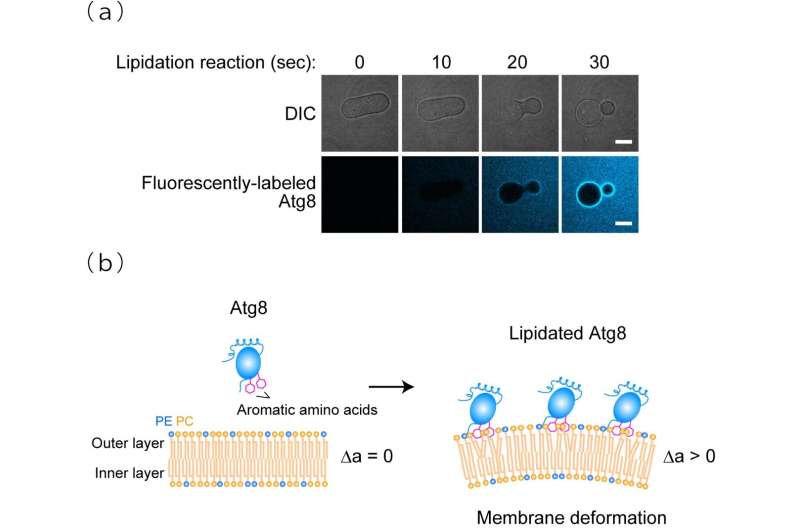
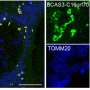
Figure 1. Membrane transforming activity of lipidated Atg8(a) Upon lipidation, prolate membranes are constricted at the hollow region and transformed into a pear-like shape. DIC, differential interference contrast. Scale bars, 10 µm. (b) The area difference between the outer and inner layers of the membrane, Δa, is increased due to the interaction of the aromatic amino acids in lipidated Atg8 with the outer layer, leading to membrane deformation. PC, phosphatidylcholine. PE, phosphatidylethanolamine. Credit: Nobuo N. Noda
Drs. Nobuo Noda (Director) and Tatsuro Maruyama (Researcher) et al. at the Institute of Microbial Chemistry (BIKAKEN, Tokyo, Japan) discovered that lipidated Atg8, the most famous factor that mediates autophagy, has membrane perturbation activity and elucidated that this activity is responsible for efficient autophagosome formation.
Autophagosome formation is an essential step in determining the target of degradation in autophagy, which is one of the mechanisms of intracellular protein degradation. It is known that lipidated Atg8 plays a primary role in autophagy processes; however, the molecular function of lipidated Atg8 on the autophagy-related membranes remains unknown.
The research group demonstrated through in vitro experiments that yeast Atg8 exhibits membrane transforming activity. Moreover, on examining the three-dimensional structure of lipidated Atg8 by solution NMR spectroscopy, they found that lipidated Atg8 interacts with the membrane via two aromatic amino acids. In addition, they found that mutant aromatic amino acids resulted in the loss of the membrane transforming activity of lipidated Atg8 in vitro, inhibited the autophagosome formation in yeast, and attenuated autophagy in mammalian cells. Consequently, they revealed a novel mechanism wherein lipidated Atg8 perturbs and transforms membranes through direct interaction, thereby promoting autophagosome formation.
The elucidation of the molecular role of the main autophagy factor, Atg8, which has been a long-standing issue in the field of autophagy, holds promise in accelerating research that will contribute to a complete understanding of the molecular mechanisms of autophagosome formation. Furthermore, it is expected to promote research and development for treating and preventing various diseases through the artificial control of autophagy by deepening our understanding of autophagosome formation mechanisms.
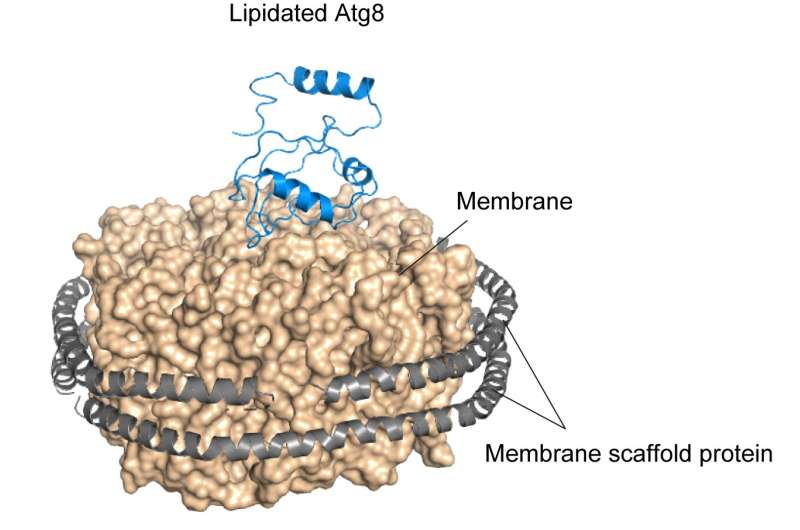
Figure 2. Structure of lipidated Atg8 Lipidated Atg8 was reconstituted on the membrane surrounded by membrane scaffold proteins, enabling us to obtain the structural information using solution NMR spectroscopy. NMR analysis showed that lipidated Atg8 has a preferred orientation on the membrane. Lipidated Atg8 and membrane scaffold protein are displayed as ribbon models. The membrane is shown as a surface model. Credit: Nobuo N. Noda
More information: Tatsuro Maruyama et al, Membrane perturbation by lipidated Atg8 underlies autophagosome biogenesis, Nature Structural & Molecular Biology (2021). DOI: 10.1038/s41594-021-00614-5
Journal information: Nature Structural & Molecular Biology
Provided by Japan Science and Technology Agency (JST)