A deeper understanding of how cells move and stick together
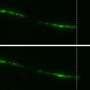
Observing how cells stick to surfaces and their motility is vitally important in the study of tissue maintenance, wound healing and even understanding how cancers progress. A new paper published in The European Physical Journal Plus, by Raj Kumar Sadhu, Weizmann Institute of Science, Rehovot, Israel, takes a step towards a deeper understanding of these processes.
"Cell adhesion is the ability of a cell to stick to another cell or an extracellular matrix. This process is important in order to understand how cells interact and coordinate their behavior in multicellular organisms," says Sadhu. "We theoretically model the adhesion of a cell-like vesicle by describing the cell as a three-dimensional vesicle adhering on a flat substrate with a constant adhesion interaction."
Alongside his co-authors, Sadhu set about exploring the role of membrane-bound curvature sensitive proteins and the forces that act on the cytoskeleton—the network of interlinking protein filaments found in the cytoplasm of all cells—during the adhesion process. The team discovered that curved proteins enhance the adhesion process considerably, especially when coupled with active cytoskeleton forces.
"Our work shows that the curved membrane proteins, coupled with the pushing force due to the cytoskeleton, can play a key role in the cell adhesion process," adds Sadhu. "In addition, we showed that these minimal ingredients are sufficient to produce a motile shape that closely resembles migrating cells. Our present work will motivate more research in this direction."
One aspect of the research that pleasantly surprised the team was the fact that the relatively simple model they created could not just describe cell adhesion, but also allowed them to capture cell movement as well. The resultant paper belongs to the topical collection "Focus Point on Mechanobiology across Scales," edited by M. Ben Amar, A. Boudaoud and M. Lenz.
"Physical principles of shape, curvature and forces, combine to give living cells their shapes," concludes Sadhu. "We show that the cells can have a variety of dynamic shapes, which spontaneously arise due to physical principles, and control the function of the cells in our bodies."
The team will seek to improve on this current study by examining the adhesion of cells on more complex surfaces. This will include curved surfaces, those with adhesion gradients, and others upon which adhesive elements are temporary.
More information: Raj Kumar Sadhu et al, Modelling cellular spreading and emergence of motility in the presence of curved membrane proteins and active cytoskeleton forces, The European Physical Journal Plus (2021). DOI: 10.1140/epjp/s13360-021-01433-9
Provided by Springer