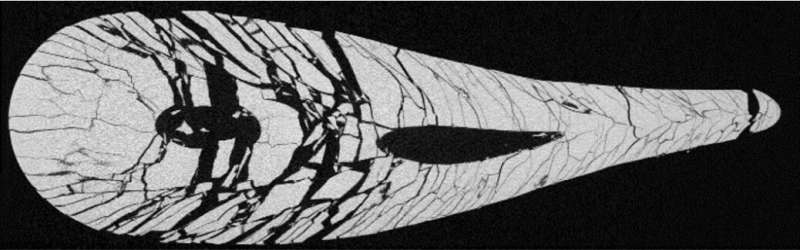
Left: a Prince Rupert’s drop. Right: when the tail of the drop is squeezed, it breaks into countless tiny fragments – all roughly the same size. Credit: S. Kooij et al., Nature Communications
When a drinking glass falls on the floor and breaks, the shards will vary in size from large to extremely small. For the broken glass of a bus shelter, the story is different: all fragments have roughly the same size. Researchers from the University of Amsterdam, Unilever Vlaardingen and EPFL Lausanne investigated the breaking phenomenon, and discovered that two very different processes cause the two types of shards. The results were published in Nature Communications this week.
The exception to the rule
The observation that there are fragments of all sizes, turns out to hold for all kinds of typical breaking and fragmentation processes. However, there are exeptions: think of broken bus shelters, where all the glass fragments have more or less the same size. This difference is not a coincidence—it turns out to be an important piece of the puzzle that leads to an explanation for the way in which normal objects break into pieces.
Stefan Kooij, Gerard van Dalen, Jean-François Molinari and Daniel Bonn inverstigated the fragmentation process, and discovered that there exist two very different ways of breaking. The special behavior of the glass in bus shelters arises because that glass has been treated in a special way, which causes the glass to have internal stresses. Those stresses eventually lead to the similarity of the fragments.
To study the process that takes place in bus shelter glass, the researchers investigated a similar kind of glass: the 'Prince Rupert's drop', also known as a 'Dutch tear'. These droplets of glass—see the left image above for an example—are made by dripping molten glass into cold water. Because the glass first solidifies on the outside and only later on the inside, causing it to shrink a bit, large stresses develop inside the drop, comparable to those in bus shelter glass. On the internet, many videos can be found displaying the special properties of the drops: they can withstand being hit with a hammer, but shatter into countless pieces when the 'tail' of the drop is pinched.
Credit: University of Amsterdam
Hierarchical and random
Using several techniques—including the use of CT-scans to measure almost 22,000 fragment sizes of a Prince Rupert's drop—the researchers studied the different fragmentation processes. By comparing the breaking of Prince Rupert's drops to the fragmentation of other materials, they were able to deduce that two types of fragmentation processes exist, which they called hierarchical and random.
When you drop a regular glass on the floor, the process that occurs is the hierarchical one. The energy contained in the motion of the glass is much larger than what is necessary to force a single fracture. To get rid of all kinetic energy, more and more fracture develop inside the glass. The process is hierarchical—it goes from large to ever smaller cracks—and results in the fact that eventually, there is no characteristic fragment size.
Credit: University of Amsterdam
By far the most situations in which objects break are of this hierarchical type. The exception arises when the energy to break the object does not come from outside, but is a result of internal stresses, as is the case in bus shelter glass and Prince Rupert's drops. In such a case, the formation of the fractures does not happen from large to small, but in a completely random way, where the 'splitting' of the cracks depends on the internal stresses. As a result, the fragments in this case do have a specific size, determined by the magnitude of the internal stress in the material.
The fact that these two types of fragmentation exist is not just an interesting curiosity; the results of the research can also lead to useful practical applications. Here, one can think of developing better safety glass for car windows, but also of the production of medicines, in which the fragmentation size plays an important role in the rate of absorption of the medicine by the body.
More information: Stefan Kooij et al. Explosive fragmentation of Prince Rupert's drops leads to well-defined fragment sizes, Nature Communications (2021). DOI: 10.1038/s41467-021-22595-1
Journal information: Nature Communications
Provided by University of Amsterdam