March 12, 2021 report
Researchers find surprising transition-metal-type bonds while building new calcium(I) complexes
A team of researchers from University Erlangen-Nürnberg, Nanjing Tech University and Philipps-Universität Marburg has found that it is possible to use calcium(I) with β-diketiminate as a ligand and potassium as a terminal reductant for dinitrogen reduction. In their paper published in the journal Science, the group describes how they tried to make a calcium(I) complex in their lab using bulky ligands as a means to stabilize the ion and how that led them to a new discovery about calcium.
Calcium is one of the most common elements in nature—in addition to being the main ingredient in bones, it is also found in seashells and some types of stone. Prior research has shown that calcium(O) is reactive, though calcium(I) is not considered to be very reactive, which means there are few if any calcium(I) complexes. In this new effort, the researchers suspected that the lack of calcium(I) complexes had more to do with lack of effort to make them than an inability to do so. They therefore set out to make one for themselves.
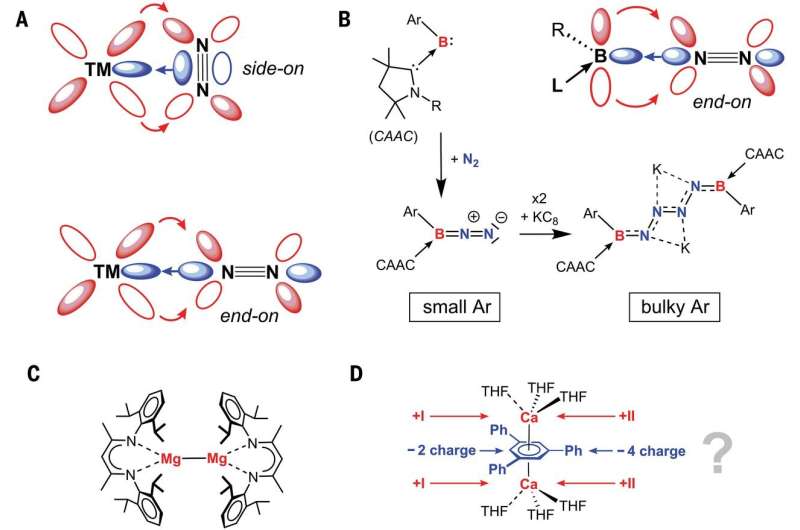
In their work, the researchers tried using β-diketiminate as a ligand to stabilize the ion. But as they did so, they found that the calcium reacted with the toluene, benzene and para-xylene they were using as solvents—that led them to try solvents that were less reactive. In so doing, they discovered that adding tetrahydropyran or tetrahydrofuran to the reaction resulted in the formation of red-brown crystals. They found that two of the calciums, which were still wrapped inside the ligand, had captured the N2 molecule that the team had used to allow for an inert atmosphere during the reaction. The team was surprised by this finding because metals such as calcium, which are known to give up electrons easily, would not have been expected to activate N2.
The team found that the calcium was able to use its d-orbital bond with N2 in ways similar to that seen with transition metals. This finding suggests chemists may have to view elements in the s-block of the periodic table in new ways.
More information: B. Rösch et al. Dinitrogen complexation and reduction at low-valent calcium, Science (2021). DOI: 10.1126/science.abf2374
Journal information: Science
© 2021 Science X Network