Chemists achieve breakthrough in the production of three-dimensional molecular structures

A major goal of organic and medicinal chemistry in recent decades has been the rapid synthesis of three-dimensional molecules for the development of new drugs. These drug candidates exhibit a variety of improved properties compared to predominantly flat molecular structures, which are reflected in clinical trials by higher efficacy and success rates. However, they could only be produced at great expense or not at all using previous methods. Chemists led by Prof. Frank Glorius (University of Münster, Germany) and his colleagues Prof. M. Kevin Brown (Indiana University Bloomington) and Prof. Kendall N. Houk (University of California, Los Angeles) have now succeeded in converting several classes of flat nitrogen-containing molecules into the desired three-dimensional structures. Using more than 100 novel examples, they were able to demonstrate the broad applicability of the process. This study will be published by Science on Friday, 26 March 2021.
Light-mediated energy transfer overcomes energy barrier
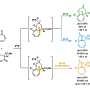
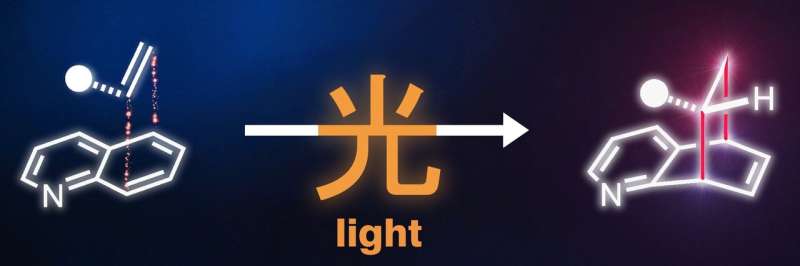
One of the most efficient methods for synthesizing three-dimensional architectures involves the addition of a molecule to another, known as cycloaddition. In this process, two new bonds and a new ring are formed between the molecules. For aromatic systems—i.e. flat and particularly stable ring compounds—this reaction was not feasible with previous methods. The energy barrier that inhibits such a cycloaddition could not be overcome even with the application of heat. For this reason, the authors of the "Science" article explored the possibility of overcoming this barrier through light-mediated energy transfer.
"The motif of using light energy to build more complex, chemical structures is also found in nature," explains Frank Glorius. "Just as plants use light in photosynthesis to synthesize sugar molecules from the simple building blocks carbon dioxide and water, we use light-mediated energy transfer to produce complex, three-dimensional target molecules from flat basic structures."
New drug candidates for pharmaceutical applications?
The scientists point to the "enormous possibilities" of the method. The novel, unconventional structural motifs presented by the team in the "Science" paper will significantly expand the range of molecules that medicinal chemists can consider in their search for new drugs: for example, basic building blocks containing nitrogen and highly relevant to pharmaceuticals, such as quinolines, isoquinolines and quinazolines, which have been scarcely used owing to selectivity and reactivity problems. Through light-mediated energy transfer, they can now be coupled with a wide range of structurally diverse alkenes to obtain novel three-dimensional drug candidates or their backbones. The chemists also demonstrated a variety of innovative transformations for the further processing of these synthesized backbones, using their expertise to pave the way for pharmaceutical applications. The method's great practicality and the availability of the required starting materials are crucial for the future use of the technology: the molecules used are commercially available at low cost or easy to produce.
"We hope that this discovery will provide new impetus in the development of novel medical agents and will also be applied and further investigated in an interdisciplinary manner," explains Jiajia Ma. Kevin Brown adds: "Our scientific breakthrough can also gain great significance in the discovery of crop protection agents and beyond."
Synergy of experimental and computational chemistry
Another special feature of the study: The scientists clarified the reaction mechanism and the exact structure of the molecules produced for the first time not only analytically and experimentally in detail, but also via computational chemistry: Kendall Houk and Shuming Chen conducted detailed computer-aided modeling of the reaction. They were able to show how these reactions work and why they occur very selectively.
"This study is a prime example of the synergy of experimental and computational theoretical chemistry," says Shuming Chen, now a professor at Oberlin College in Ohio.
"Our detailed mechanistic elucidation and understanding of reactivity concepts will enable scientists to develop complementary methods and to use what we learned to design more efficient synthetic routes in the future," adds Kendall Houk.
The story behind the publication
Using the method of light-mediated energy transfer, both Jiajia Ma/Frank Glorius (University of Münster) and Renyu Guo/Kevin Brown (Indiana University) had success, independently. Through collaborations with Kendall Houk and Shuming Chen at UCLA, both research groups learned of the mutual discovery. The three groups decided to develop their findings further together in order to share their breakthrough with the scientific community as soon as possible and to provide medicinal chemists with this technology to develop novel drugs.
More information: Jiajia Ma et al. Photochemical intermolecular dearomative cycloaddition of bicyclic azaarenes with alkenes, Science (2021). DOI: 10.1126/science.abg0720
Journal information: Science
Provided by University of Münster