Biosensing with whispering-gallery mode lasers
Whispering-gallery-mode (WGM) microresonators are opening up many new research directions that enable the detection of proteins, enzymes and DNA, down to single molecules. A new class of sensors makes use of active microresonators to drastically expand the functionality and range of WGM sensor applications, especially for biological and chemical assays. We review the most recent advances of WGM microlasers for biosensing and provide an outlook on exciting new research directions and emerging WGM sensor applications.
Label-free optical sensors based on optical whispering-gallery-mode (WGM) microresonators exhibit extraordinary sensitivity for detecting physical, chemical, and biological entities, even down to single molecules. This advancement in label-free optical detection is made possible by application of the optical microresonator, i.e. a 100 um glass microspheres, as optical cavity to enhance the detection signal. Akin to a spherical micromirror, the WGM cavity reflects the light by near-total internal reflection and thereby creates multiple cavity passes that enhance the optical detection of analyte molecules interacting with the evanescent field.
In contrast to the 'cold' WGM microresonators, the emerging active WGM microlasers have the potential to significantly expand the number of possible applications of this class of sensors in biological and chemical sensing, and especially in in vivo sensing. The WGM microlasers can sense from within tissue, organisms and single cells, and they can be used to improve upon the already impressive single-molecule detection limits of the 'cold'-cavity optoplasmonic WGM sensors.
Here, we review the most recent advances of WGM microlasers in biosensing. In contrast to the 'cold' cavity WGM sensors, the active WGM microresonators make use of gain media such as dye molecules and quantum dots to compensate for optical loss and to achieve lasing of the WGM modes. Similar to other conventional lasers, lasing is observed from narrow spectral lines in the WGM emission spectra.
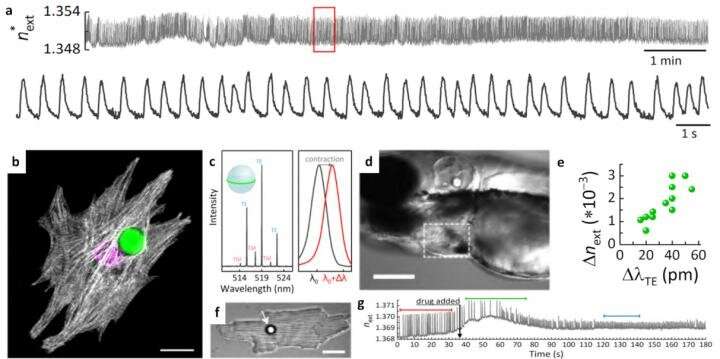
We review the main building blocks of WGM microlasers, recently demonstrated sensing mechanisms, the methods for integrating gain media in WGM sensors, and the prospects for active WGM sensors to become a useful technology in real-world applications. We review WGM microlaser sensing experiments at the molecular-level where lasing spectra are analyzed to study the binding of molecules, to sensing at the cellular level where microlasers are embedded into or integrated with single cells to enable novel in vivo sensing and single-cell tracking applications (see figure).
More information: Nikita Toropov et al, Review of biosensing with whispering-gallery mode lasers, Light: Science & Applications (2021). DOI: 10.1038/s41377-021-00471-3
Journal information: Light: Science & Applications
Provided by Chinese Academy of Sciences