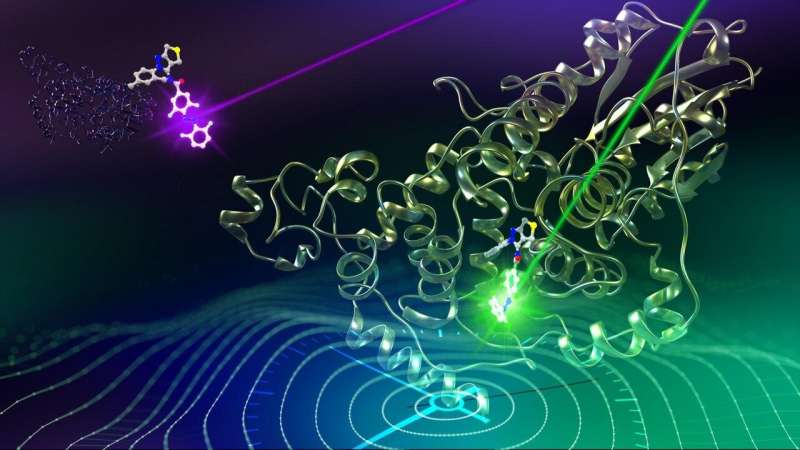
Trans and cis isomers of azobenzene determine the change in the circadian period through their interaction with the CRY1 clock gene Credit: Issey Takahashi
The Nagoya University Institute of Transformative Bio-Molecules (WPI-ITbM) research team of Designated Associate Professor Tsuyoshi Hirota, Postdoctoral Fellow Simon Miller, Professor Kenichiro Itami and graduate student Tsuyoshi Oshima (Research Fellowship for Young Scientists, JSPS), in collaboration with the group of Professor Ben Feringa and Postdoctoral Fellow Dušan Kolarski of Groningen University in the Netherlands, have achieved a world first: fully reversible manipulation of the period of the circadian clock using light, by exchanging part of a compound with a light-activated switch.
Waking in the morning, sleeping at night—the majority of our biological activities repeat within a daily cycle. The internal process which governs this rhythm is known as the circadian clock. While it is understood that the circadian clock is controlled by the combined functions of clock genes and clock proteins, the process by which it is possible to control and stabilize the rhythm over the lengthy period of a day has been shrouded in mystery. In order to tackle this question, the researchers established a chemical biology process for large-scale analysis of the effect of compounds on the circadian rhythm in cultured human cells, elucidating the significant molecular mechanisms which determine the daily period.
This large-scale chemical screening identified two compounds—TH303 and its analogue TH129—which lengthened the circadian clock period. The research team then worked on elucidating how these compounds interact with the clock protein CRY1 at a molecular level using X-ray crystallography. They found that part of these compounds, known as a benzophenone, possessed a similar structure to the cis isomer of azobenzene, a light-activated switch. When they then analyzed the response to light of GO1323, a variant of TH129 in which benzophenone is displaced by azobenzene, they found that its structure changed to the cis isomer under ultraviolet light, and back to the trans isomer under white light. According to computer simulations, the cis isomer of GO1323 interacts identically to TH129 with CRY1, while the trans isomer has no interaction with it.
Thus, when exposed to ultraviolet light, the circadian clock period of cultured human cells which had been treated with GO1323 was extended compared with those which had been kept in the dark. Furthermore, when exposed to white light, these cells' circadian clock period returned to normal, proving that the process is reversible. As ultraviolet light is damaging to cells, the research team had to find a way to adapt the process to use a non-harmful area of the spectrum to extend the period. They synthesized GO1423, containing tetraorthofluoroazobenzine. This compound changes to its cis isomer under green light, and to its trans isomer under violet light, while maintaining the other desirable characteristics of GO1323. When cells treated with GO1423 were exposed to green light, their circadian rhythm period was extended compared with those which had been kept in the dark, and when exposed to violet light, the effect was reversed. Thus, the researchers succeeded in producing a reversible method for controlling the circadian clock period using visible light.
Control of the circadian clock using methods such as these is expected to contribute to the treatment of related diseases such as sleep disorders, metabolic syndrome and cancer, and this research achievement represents an important and exciting step forward in the field.
More information: Dušan Kolarski et al, Photopharmacological Manipulation of Mammalian CRY1 for Regulation of the Circadian Clock, Journal of the American Chemical Society (2021). DOI: 10.1021/jacs.0c12280
Journal information: Journal of the American Chemical Society