How maize makes an antibiotic cocktail

Maize (Zea mays) produces a plethora of antibiotics called zealexins. Even though scientists have identified at least 15 zealexins, they suspect there are even more to find. Zealexins are produced in every corn variety and protect maize by fending off fungal and microbial infections. Scientists had assumed that maize synthesizes these compounds via clear, straight pathways, entailing the function of more than a dozen enzymes. But researchers have now revealed that just a handful of enzymes are needed to transform zealexins and related precursors into a bonanza of diverse structures.
Zealexins belong to a class of chemicals called terpenoids, which have a structure belonging to a group of chemicals called terpenes. Terpenes are important in plant growth, defense, and environmental interactions, and also have industrial applications, including bioenergy. All plants make terpenoids, which carry out a diverse array of functions, including acting as antibiotics. But zealexins are almost completely limited to maize. By deciphering the zealexin synthesis pathway, this study makes it possible to harness the protective function of zealexins and related compounds for other plants, including those cultivated for bioenergy. The zealexin synthesis pathway may be especially amenable to transfer into other plants because some of its enzymes can transform diverse terpene substrates into many products; giving scientists a bigger bang for their buck. The research team reports that transferring just four genes encoding these enzymes to another plant, such as Sorghum bicolor—a candidate bioenergy feedstock and JGI Flagship Plant—could produce at least 15 zealexins. Moreover, zealexin biosynthetic enzymes would also act on native substrates to produce additional novel antibiotics.
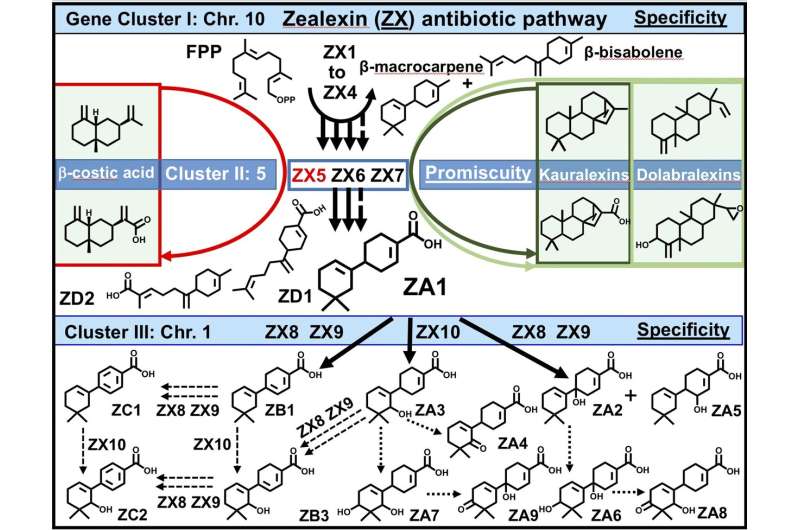
The zealexins are chemically complex, but their biosynthetic pathway is simpler than scientists had suspected. A team recently reported in Nature Plants that the zealexin pathway has an "hourglass" shape with diverse precursors in, and many different products out, instead of the more typical, one enzyme, one product. At each step, the zealexin pathway utilizes dexterous enzymes— terpene synthases and cytochrome P450s—that catalyze multiple products, resulting in many antibiotics.
To uncover the enzymes, the team used a battery of techniques. They screened panels of hundreds of maize lines for genetic variation in the ability make zealexins, and harnessed gene expression data from these lines to identify genes associated with zealexin synthesis. The researchers used proteomics to home in on precise biosynthetic enzymes elicited by fungal infection. They analyzed differences among mutant maize strains, deploying gene editing to knock out four critical zealexin pathway genes. And they transferred genes of interest to tobacco (Nicotiana benthamiana), giving the plant the ability to produce diverse zealexins. These genes were synthesized by the U.S. Department of Energy (DOE) Joint Genome Institute (JGI), a DOE Office of Science User Facility, through its Community Science Program. And the work is part of a larger CSP Functional Genomics proposal to understand terpene synthesis and function across many plants in addition to maize, including the bioenergy crop switchgrass.
The team confirmed just how important zealexins are to maize health. Infections with a stalk-rotting fungus (Fusarium graminearum) and a bacterium that causes wilting (Pantoea stewartii) were much more severe when the zealexin biosynthetic pathway was knocked out.
Interestingly, the enzymes the team uncovered were found in multiple, semi-redundant clusters on different maize chromosomes. The collective results demonstrate that the zealexin pathway is a critical biochemical defense in maize, which has been largely overlooked, according to the study's senior author Alisa Huffaker, a biologist at the University of California, San Diego. The findings could help scientists produce more robust bioenergy plants in the future.
More information: Yezhang Ding et al. Genetic elucidation of interconnected antibiotic pathways mediating maize innate immunity, Nature Plants (2020). DOI: 10.1038/s41477-020-00787-9
Journal information: Nature Plants
Provided by DOE/Joint Genome Institute