Controlling lasers with dancing DNA

DNA is the hereditary material in the nucleus of all cells in humans and other living organisms. Besides its significance in biology, DNA has also played a specific role in controlling many physical devices. Recently, an international research team at Nanyang Technological University, Singapore, has demonstrated the concept of a switchable microlaser by taking advantage of the organic biomolecule DNA hybridization process.
To date, advances in switchable microlasers have emerged as a building block with immense potential in controlling light-matter interactions and integrated photonics. Generally, optical switching is achieved by complex device fabrication or some physical approaches, such as modifying the structure or refractive index of the lasing cavities. Contrary to artificially-designed interface, stimuli-responsive biointerfaces take advantage of a biological system and bio-recognition such that a higher level of functionalities could be realized at the nanoscale. Nonetheless, switching laser emission with biological recognition has yet to be addressed, particularly with reversible and wavelength tunability over a broad spectral range.
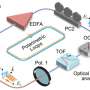
To address this issue, Chen's team developed a novel method to switch laser emission by incorporating DNA in an optical microcavity. DNA is one of the most potent biomaterials known for its controllable synthesis and specificity of base-pair interactions. The programmability and self-assembly of DNA structures offer versatile ways for constructing DNA biointerfaces and tailoring optical response. The Fabry-Perot optical microcavity consists of two dielectric mirrors, in which dye-doped liquid crystals were introduced as optical gain to enhance the response of DNA binding events.
The strong light-matter interaction induced by the microcavity thus enables subtle changes to be amplified within the cavity and liquid crystal matrixes. The liquid crystal molecule changes from homeotropic to planar alignment when single-stranded DNA (sDNA) is adsorbed on the cationic monolayer of the matrix. The orientation changes of LC molecules thus resulted in a blue-shift of lasing wavelength with pronounced signal amplification. The lasing wavelength could be reverted upon binding with its complementary part through DNA hybridization process.
"We used this special DNA-liquid crystal interaction as the switching power to alter the liquid crystals orientation in the Fabry-Perot microcavity so that laser emission switching among different wavelengths was achieved," said Professor Yu-Cheng Chen, the study's corresponding author. The interactions lead to temporal switching of lasing wavelengths and intensities. The lasing wavelength appears blue shift when ssDNA introduced. It reverts upon hybridizing with its complementary bases. Both experimental and theoretical studies revealed that absorption strength of the gain medium is the critical mechanism that determines the laser shifting behavior.
"The significance of this study is to introduce the concept of using organic biomolecules to switch coherent light sources at different wavelengths. It represents a milestone in achieving biological-controlled laser," said Chen. The team believes that this study sheds light on the development of programmable photonic devices at the sub-nanoscale by exploiting the complexity and self-recognition of biomolecules. By exploiting the complexity and self-recognition of DNA sequences, laser light could be fully manipulated and programmed. The remarkable ability of specific molecular recognition could be potentially suitable for applications such as information encoding and data storage with laser light in the future. This work was published in ACS Nano.
More information: Yifan Zhang et al. DNA Self-Switchable Microlaser, ACS Nano (2020). DOI: 10.1021/acsnano.0c08219
Journal information: ACS Nano
Provided by Nanyang Technological University