Novel cellulose finding may lead to new chemicals, biofuels
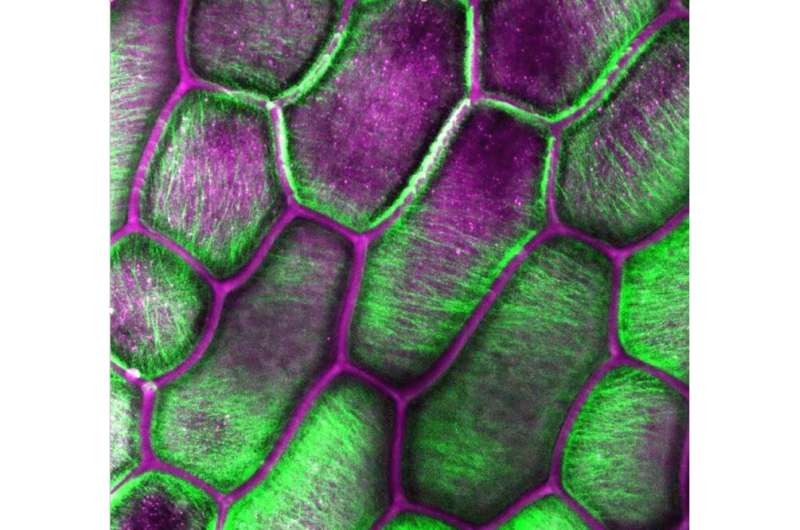
A multi-institutional research group led by two Penn State faculty members has identified, for the first time, how cellulose crystals orient themselves relative to the cell wall in plants, with potential implications for chemical and energy development.
"A more detailed understanding of the structure of plant cell walls could lead to new strategies for breakdown of these materials to create fuels and high-value chemicals," said Enrique Gomez, co-principal investigator of the study and professor of chemical engineering and materials science and engineering with a co-appointment in the Materials Research Institute. "As such, our work may lead to new strategies for biofuel or biochemical production."
Cellulose is the most abundant biopolymer on earth. It enables plants to build stems, trunks and leaves. It also protects sugars used by plants for various life functions. To convert cellulosic biomass into new biofuels and biochemicals, researchers need to understand how to break down cellulose. Cellulose is difficult to degrade into raw materials for the manufacturing of biofuels and biochemicals.
The versatile, energy-rich material has long been known to be crystalline, but there are still some mysteries about its structure and how it forms. According to Gomez, researchers have speculated whether cellulose crystals twist for years. The Penn State team found that the crystals have a "preferred orientation," which is a tendency to be positioned in one way without twisting. These findings were published in September in Nature Communications.
"Our work identified a new type of organization in plant cell walls, in that we identified that crystals within plant cell walls have a preferred orientation," said Esther Gomez, co-principal investigator of the study and associate professor of chemical engineering and biomedical engineering. "This is a surprising result, which we found in three different plant species, suggesting our finding is due to some common consequence of how plants make their cell walls. Along the way, we may have helped settle a long-standing debate—whether crystals within plant cell walls twist—because the preferred orientation suggests that crystals don't twist. If they did, there wouldn't be a preferred orientation."
To "see" cellulose's crystalline orientation in the plant samples, they used a technique not previously applied, to their knowledge, in the study of plant walls.
"The technique, grazing-incidence wide-angle X-ray scattering (GIWAXS), was developed for materials science and used extensively for the study of thin films, including polymer films," Esther Gomez said.
GIWAXS involves an X-ray beam hitting a thin film sample, in this case cellulose, at very shallow angles. This can be adjusted to probe only the surface or bulk of the sample at molecular length scales. The plant species they studied included the crystalline structures in the cell walls of onions, thale cress and moss.
This discovery could lead to the opening of new avenues of cellulose research, especially in bioenergy and biochemicals, according to the researchers.
"Allowing the study of this new type of organization will lead to new fundamental studies on how the remarkable properties of cell walls arise," Enrique Gomez said.
More information: Dan Ye et al. Preferred crystallographic orientation of cellulose in plant primary cell walls, Nature Communications (2020). DOI: 10.1038/s41467-020-18449-x
Journal information: Nature Communications
Provided by Pennsylvania State University