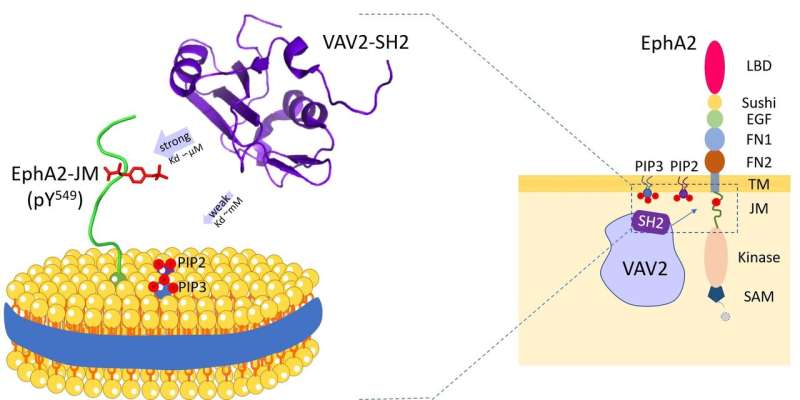
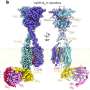
Vav2 protein binds to cell membrane phospholipids and the receptor tyrosine kinase EphA2 juxtamembrane region. Credit: HFIPS
Recently, a research team led by Prof. Wang Junfeng from the High Magnetic Field Laboratory, Hefei Institutes of Physical Science revealed the dual specificity of Vav2-SH2 protein after investigating the specific recognition mechanism of human Vav2 protein with cell membrane phospholipids and the receptor tyrosine kinase EphA2 juxtamembrane region.
Vav2 is broadly expressed in human tissues and plays essential roles in various biological processes. And through its Src-homology 2 (SH2) domain, Vav2 is able to interact with several transmembrane receptors. These SH2-mediated interactions take place on or near the plasma membrane and are essential for Vav2 to mediate different extracellular signal transduction.
In this research, with the help of nuclear magnetic resonance (NMR) techniques, the researchers discovered the role of the phospholipid bilayer environment on protein-protein interaction, offering a potential method to study this interaction in the same environment under near physiological conditions.
The researchers revealed that the Vav2-SH2 domain specifically binded to the Y594 phosphorylated juxtamembrane region using its conserved protein binding site. Interestingly, it also had the phospholipid binding ability and the phospholipid binding site of Vav2-SH2, adjacent to the protein binding site, was determined by NMR experiments.
Furthermore, the researchers used lipid nanodiscs to mimic the membrane, and uncovered the role of the membrane environment in modulating this protein-protein recognition.
More information: Liang Ge et al. Biochemical and NMR characterization of the interactions of Vav2–SH2 domain with lipids and the EphA2 juxtamembrane region on membrane, Biochemical Journal (2020). DOI: 10.1042/BCJ20200300
Journal information: Biochemical Journal
Provided by Chinese Academy of Sciences