Engineered electrode material moves battery development closer to fast charging

Electric vehicles are gaining in popularity, but their long charging time is a significant detraction for potential customers. While a typical SUV with a combustion engine could travel 300 miles with a five-minute refuel, a state-of-the-art electric vehicle takes about one hour to store enough energy to travel the same distance. The technology for a high-capacity lithium-ion battery that charges quickly and operates efficiently is still an unrealized goal—but researchers are now closer than ever.
An international team of researchers published details of an engineered electrode material that allows for such advanced batteries on Oct 8th in Science.
"The combination of high energy, high rate, and long cycle life is the holy grail of battery research, which is determined by one of the key components of the battery: the electrode materials," said Hengxing Ji, professor at the University of Science and Technology of China (USTC). "We aim to search for an electrode material that can make a dent in performance metrics from laboratory research and can hold the promise to stand with the industrial production techniques and requirements."
Energy enters and leaves the battery by electrochemical reactions in electrodes, so efficient and effective lithium-ion transfer is of the utmost importance, according to first author Hongchang Jin of USTC, especially in transferring the energy from the battery to the device via the anode.
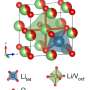
The researchers turned to black phosphorus, a material that has been considered for use in electrodes before but is usually abandoned due to its tendency to deform along its layered edges, making the transfer of lithium-ions deeply inefficient and rendering a lower quality material. By combining black phosphorus with graphite, the chemical bonds between these two materials stabilize and prevent the problematic edge changes.
The team also tackled another issue hindering the material: Electrolytes can break down into less conductive pieces and build up on the surface of the electrode, inhibiting lithium-ion transfer into the electrode material, like dust obscuring light through glass. The team applied a thin polymer gel coating to the electrode materials and reinforced the lithium-ion transport path, effectively preventing the issue.
"The composite anode material restored 80% of its full capacity in less than 10 minutes and shows a 2000-cycle operation life at room temperature, which was measured at conditions compatible with the industrial fabrication processes," said co-first author Sen Xin, professor of the Institute of Chemistry Chinese Academy of Sciences. "If scalable production can be achieved, this material may provide an alternative, updated graphite anode, and move us toward a lithium-ion battery with energy density of more than 350 watts-hour per kilogram and fast-charging capability. Successful projection of the above parameters onto the electric vehicle will significantly raise its competitiveness against the fuel cars."
The 350 watts-hour per kilogram describes the energy capacity of the battery—an electric vehicle with such a battery could travel 600 miles on a single charge. For comparison, the on-market Tesla Model S can travel 400 miles on one charge.
With this novel technology, Ji said the researchers plan to pursue both fundamental scientific questions of the lithium-ion charging-discharging process and industry-related questions on ways to scale composite material production in more mild conditions.
"We will investigate engineering materials of rationally selected structure, but with consideration for price and practicality to achieve an attractive performance," Ji said.
More information: Black phosphorus composites with engineered interfaces for high-rate high-capacity lithium storage. Science (2020). science.sciencemag.org/cgi/doi … 1126/science.aav5842
Journal information: Science
Provided by University of Science and Technology of China