Cholesterol binding sends long-distance communication signals in proteins
Humans with a high cholesterol fear the "bad cholesterol"—the so-called low-density lipoprotein (LDL)—because it is genetic and cannot be regulated with medication. However, a healthy occurrence of LDL is important for cellular processes. LDL takes up cholesterol esters—which contain both cholesterol and fatty acid—in the blood and transports it to the cell. In the vesicular lysosomes of the cell, the ester is broken down by enzymes. The resulting free cholesterol is then transported to other parts of the cell, such as the endoplasmic reticulum and other cell membranes, where it is needed for cell processes. A cholesterol balance (homeostasis) is created in the lysosomes.
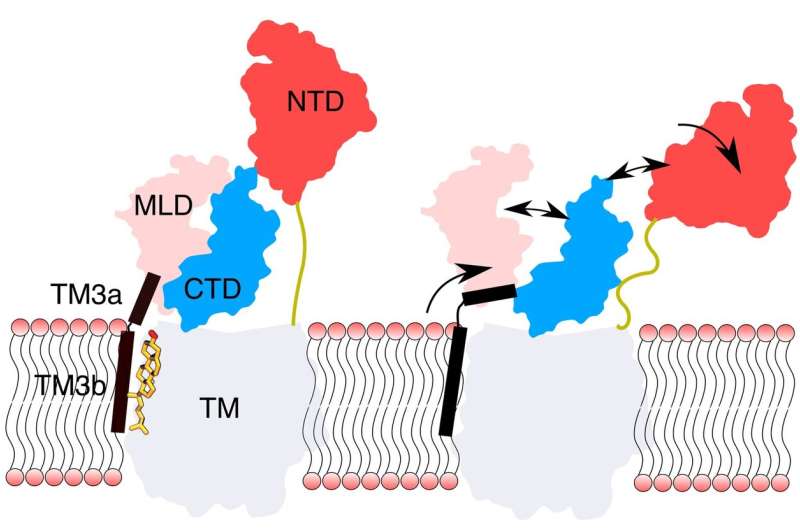
The protein NPC1, which sits in the lysosomal membrane, seems to play an important role in cholesterol transport in the cell. It has a so-called sterol-sensing domain (SSD) in the membrane and a region known as the N-terminal domain (NTD) inside the lysosome. Based on current knowledge, the SSDs should regulate the activity of NPC1 by binding cholesterol.
Serious diseases caused by disturbed cholesterol transport
It is known that a specific pathological genetic modification of the protein NPC1 causes cholesterol esters to accumulate in the lysosomes rather than break down into free cholesterol. The disease is known as Niemann Pick type C disease, and it affects the liver and spleen, ultimately leading to neurological damage. However, the exact mechanisms leading to the accumulation of cholesterol esters are not yet understood, so a research group led by Himanshu Khandelia, Associate Professor at the University of Southern Denmark, explored the underlying processes with simulations run on the "Piz Daint" supercomputer at CSCS. In the process of studying how membrane proteins behave in the presence and absence of free cholesterol, they made a surprising discovery.
The atomistic simulations showed that, without cholesterol in the sterol-sensing domain (SSD), there are central effects on the N-terminal domain (NTD) of the protein NPC1, which is eight nanometres away. Specifically, without the binding of cholesterol to the SSD of the lysosomal membrane, the luminal region of the NPC1 protein becomes highly mobile. This causes the NTD to move laterally away and bend towards the membrane where it loses contact with the luminal area. The researchers suspect that the NTD area tries to reach free cholesterol of another protein (NPC2) by bending. Khandelia emphasizes that it is not usual in simulations to observe such long-distance molecular processes.
Cholesterol binding as a regulator
According to their study recently published in PLoS Computational Biology, the researchers say the results of the simulations suggest that cholesterol bound to the SSD acts as a conformational brake, i.e. it prevents the spatial arrangement of the protein from changing. Under the binding of cholesterol, the NTD assumes an upright position, which then allows the cholesterol to pass through the "protein tunnel" to the membrane, from where it can be transported to other cellular compartments.
Even though the simulation took place under idealized conditions, the researchers assume that it provides basic insights into the functioning of the NPC1 protein.
"This could lead to better drug treatment of Niemann-Pick disease in the future," Khandelia points out.
More information: Vikas Dubey et al. Cholesterol binding to the sterol-sensing region of Niemann Pick C1 protein confines dynamics of its N-terminal domain, PLOS Computational Biology (2020). DOI: 10.1371/journal.pcbi.1007554
Provided by Swiss National Supercomputing Centre