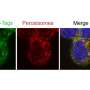
View into plant cells: A membrane protein is targeted to two locations
Metabolic processes are especially complex in plants due to their obligate sessile lifestyle, and scientists are discovering new and surprising connections that occur within plant cells. An important metabolic route that has occupied plant scientists for decades is the so-called oxidative pentose-phosphate pathway by which carbohydrates are converted to reduction power. For this pathway, two membrane proteins play an important role—GPT1 and GPT2. They import activated glucose in the form of glucose-6-phosphate into plastids, special cell organelles of plants, to feed the oxidative pentose-phosphate pathway. This process is also important for plant reproduction, especially during pollen, ovule and seed development.
Since there were indications that the three oxidative steps of the pathway may also occur in other cell organelles, the peroxisomes, researchers from the University of Münster (Germany) wondered whether membrane proteins were also directed simultaneously to two different sites within the same cell. Together with colleagues at the University of Düsseldorf (Germany) they found an answer. Their results show that only GPT1 is distributed to two locations—and the alternative route of the membrane protein leads via the endoplasmic reticulum, a net-like organelle of the cell. Thus, GPT1 enables the formation of reduction power, i.e. the capability to transfer electrons, at two sites simultaneously.
"Our study shows that besides plastids and the cytoplasm, the oxidative pentose-phosphate pathway is also a main source of the energy-rich coenzyme NADPH in peroxisomes," says study lead Prof. Antje von Schaewen from Münster University. The researchers suppose that plants in which this pathway is blocked in peroxisomes may be less stress-resistant. The study was published in the journal The Plant Cell.
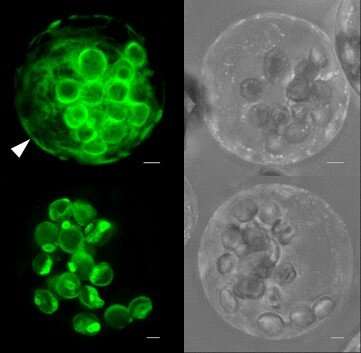
The scientists investigated the processes in Arabidopsis thaliana, a genetic model plant. In previous studies with this species, they had already shown that other enzymes of the pathway are redirected from plastids or the cytoplasm to peroxisomes under certain conditions. For this purpose, they employed fluorescent reporter fusion proteins. This method was also used in their current study to visualize GPT1 and GPT2 by life-cell imaging and modern light microscopic techniques.
They found that only GPT1 targets both plastids and the endoplasmic reticulum, from which new peroxisomes are formed in a particular region. The scientists observed that a still unknown factor initially prevents the transport of GPT1 to peroxisomes—until the membrane protein is needed there. "By experimentally enforcing interaction with peroxisomal import proteins at the endoplasmic reticulum, we could show that GPT1 can be 'dragged' to its alternative location," explains first author Dr. Marie-Christin Baune.
After the researchers had discovered that GPT1 occurs at peroxisomal membranes, they investigated which transport processes may occur in an artificially reconstituted system from yeast. They found that GPT1 prefers to exchange glucose-6-phosphate for ribulose-5-phosphate, the product that leaves the organelle. Ribulose-5-phosphate is an important precursor of nucleotides, the building blocks of nucleic acids.
Immunoblot analyses, by which proteins are visualized, additionally suggested that GPT1 is not only essential at plastids during fertilization but also at peroxisomes. Thus, GPT1 differs markedly from its "brother" GPT2—a result that the scientists had not expected, and which also became evident in genetically modified plants.
"Our results suggest that GPT1 and GPT2, despite their high similarity, fulfill only minimally overlapping tasks in plants. The loss of GPT2 is tolerated, at least under laboratory conditions. GPT1 however is indispensable, both at plastids and peroxisomes," says co-author Dr. Hannes Lansing.
"Since we now know that all reactions of the oxidative pentose-phosphate pathway that produce NADPH and ribulose-5-phosphate may occur in peroxisomes, we want to find out in future studies which other processes depend on them," says Antje von Schaewen.
More information: Marie-Christin Baune et al, The Arabidopsis Plastidial Glucose-6-Phosphate Transporter GPT1 is Dually Targeted to Peroxisomes via the Endoplasmic Reticulum, The Plant Cell (2020). DOI: 10.1105/tpc.19.00959
Journal information: Plant Cell
Provided by University of Münster