Support drives fate of protected gold nanoclusters as catalysts

In collaboration with experimentalists from Ghent University, Belgium and Utrecht University, Netherlands, researchers at the Nanoscience Center (NSC) at the University of Jyväskylä, have recently discovered that the choice of a support material for model catalysts, made from gold nanoclusters protected by organic molecules, may have drastic effects on the structure of the catalyst. On certain supports, the clusters completely disintegrate, while on others, the organic protective layer peels away leaving behind the intact metallic nanoclusters that can act as catalysts for a desired reaction. The research was published in Chemistry-A European Journal (2020).
Catalysts are important to produce chemicals used in our daily lives. They save a lot of energy and make chemical reactions faster as compared to their uncatalysed counterparts.
Nanomaterials, especially metallic nanoclusters are widely used owing to their high efficiency and are generally placed on an inactive oxide support for these applications. However, these nanoclusters are at times less stable, and are protected hence with a layer of organic molecules. The present study is an important step towards the design, control, and synthesis of atomically precise supported catalysts with tailored physical and chemical properties.
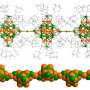
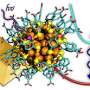
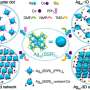
Gold nanoclusters (Aun) of different sizes protected by phosphine organic molecules were deposited on four different supports and their properties were measured using X-ray absorption spectroscopy. On Bronsted acid supports (surfaces that have the tendency to give away protons), the clusters were completely fragmented breaking up the Au cluster, while on Lewis acid supports (surfaces that have the tendency to gain electrons) the phosphine organic layer peeled away leaving the metallic Aun cluster preserving the original size of the cluster.
The theoretical models developed in Jyväskylä explained the experimental observations by studying the charge transfer between the support and the clusters.
This study was published in the international publication series Chemistry-A European Journal and recognized as a "hot" paper. An image describing the work was also selected as a cover feature in the journal's recent issue of June 2, 2020.
In Jyväskylä, post doctoral researcher Nisha Mammen, Professor Karoliina Honkala, and Academy Professor Hannu Häkkinen were responsible for the theoretical part of the work. The research was supported by the Academy of Finland. The computer simulations in the study were carried out in the local university supercomputers as well as those of the CSC—IT Center for Science.
More information: Alessandro Longo et al, Towards Atomically Precise Supported Catalysts from Monolayer‐Protected Clusters: The Critical Role of the Support, Chemistry – A European Journal (2020). DOI: 10.1002/chem.202000637
Journal information: Chemistry – A European Journal
Provided by University of Jyväskylä