Adsorbent material developed from PET bottles for the removal of antibiotics from water
South Korea, with its high antibiotic use, is categorized as a country at high risk of the emergence of multi-drug-resistant bacteria, or so-called "super bacteria." According to the Ministry of Environment, antibiotic substances have been detected at livestock wastewater treatment facilities, sewage treatment plants and in rivers.
The Korea Institute of Science and Technology announced that a research team, led by researchers Jung Kyung-won and Choi Jae-woo, at KIST's Water Cycle Research Center, has developed a high-efficiency, adsorbent material using PET waste bottles. The new material is expected to help solve the problem of environmental toxins and antibiotic-resistant bacteria which are caused by leaks of antibiotics into water.
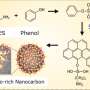
Currently, the most well-known method of effectively removing antibiotics from water uses porous carbon composite, synthesized by pyrolyzing metal-organic frameworks (MOF). Porous carbon composites adsorb antibiotics in the water, thereby removing them. However, since the organic ligand generally used to synthesize MOF is very expensive, the cost is a major obstacle to this method's widespread, practical application through mass production.
In order to develop a more cost-effective solution, the KIST research team turned its attention to the PET bottles that people use in their everyday lives. PET is a high-molecular compound obtained by polymerizing ethylene glycol and terephthalic acid, the latter of which is used as organic ligand for the syntheses of MOF. The KIST research team extracted high-purity organic ligand from PET waste bottles and used it to synthesize a high-efficiency adsorbent material that could effectively remove antibiotics from water in an environmentally and economically beneficial way.

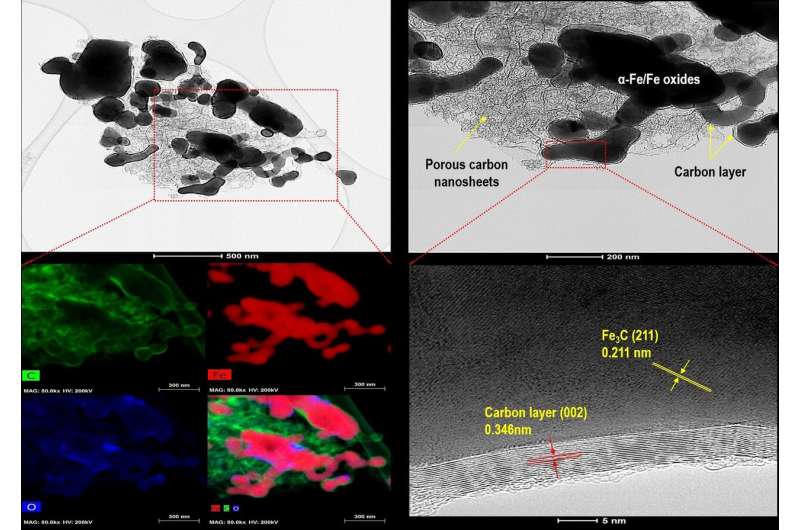
During the development of this adsorbent material, an alkaline hydrolysis process was used to induce a neutralization reaction, resulting in the production of a high-purity terephthalic acid. To maximize the efficiency of the alkaline hydrolysis process, the research team incorporated an ultrasound-assisted phase transfer catalyst process. By optimizing this process, the team was able to successfully extract 100% high-purity terephthalic acid, which they then used to develop a porous carbon composite. Iron-based MOF was used as a precursor in order to impart magnetism to the adsorbent material. In this way, the team was able to develop an eco-material that can be easily separated from the mixture after the adsorption process, using an external magnetic field.
The KIST research team tested the efficiency of the porous carbon composite in terms of its ability to adsorb "tetracycline," or the antibiotic used to treat bacterial infections, from the water. Tests showed that the newly developed material was able to remove 100% of the tetracycline in about 90 minutes under general water conditions (pH 6), with an adsorption rate of 671.14 mg/g, which is a rate superior to that of previously developed adsorbents. In order to assess the reusability of the porous carbon composite, the adsorption-desorption process was conducted five times. Even after repeated use, the material maintained 90% of its adsorption properties, indicating a high degree of stability and wide applicability for water treatment.
Dr. Jung Kyung-won at KIST said, "This porous carbon composite is applicable to a wide range of water treatment areas as it uses waste plastics to prevent environmental pollution and maintains its high adsorption properties even after repeated use."
KIST's Dr. Choi Jae-woo said, "The porous carbon composite developed through this research is applicable to various fields, ranging from eco-materials to energy materials, and I expect that it will soon be highly regarded as a value-added eco-material."
More information: Ju‐Myung Kim et al, Ecofriendly Chemical Activation of Overlithiated Layered Oxides by DNA‐Wrapped Carbon Nanotubes, Advanced Energy Materials (2020). DOI: 10.1002/aenm.201903658
Journal information: Advanced Energy Materials
Provided by National Research Council of Science & Technology