February 24, 2020 feature
Programmable droplet manipulation by a magnetic-actuation robot

Droplet manipulation in materials science can contribute to water collection, medical diagnostics and drug delivery techniques. While structure-based liquid operations are widely used in nature and in bioinspired artificial materials, laboratory strategies depend on fixed structures for unidirectional water movement. In a new study on Science Advances, An Li and a research team in the Institute of Chemistry and the University of Chinese Academy of Sciences in China proposed to develop a magnetic-actuated robot. The construct had adjustable structures to distribute forces of resistance and determine droplet behavior. The robot could transport, split, release and rotate droplets for universal applications in various fields and rough environments. The new findings offer an efficient strategy for automated droplet manipulation.
In this work, Li et al. used reliable methods to manipulate droplets using a magnetic-actuated robot with steel beads in a programmable magnetic field. The scientists controlled the structure of the robot by adjusting the magnetic field. The differentiated robot structures led to diverse droplet behavioral combinations including split, release, rotation and transport. The robot is universally applicable to various fluids including water, oil and gas. Li et al. aided robot actuation across limited spaces, on uneven surfaces and even under anhydrous or anaerobic conditions. The work has great potential for material transportation, microfabrication and in clinical medicine.
In-lab droplet manipulation is inspired by nature. For example, plants and animals have displayed fog collection systems in cacti, water collection on spider silk and influenced the behavior of the Namib Desert beetle to collect and move water. The asymmetric and intrinsic structure for water sample collection can induce a Laplace pressure gradient in the droplets, causing motion in a preset direction. Researchers can use various external strategies including electricity, magnets, acoustics and wetting surfaces to actuate their motion. Of these, magnetic force has the advantage of long-range action, safety and ease-of-control. Researchers had used magnetic particles to drag water droplets across hydrophobic or patterned surfaces and across magnetically responsive materials.
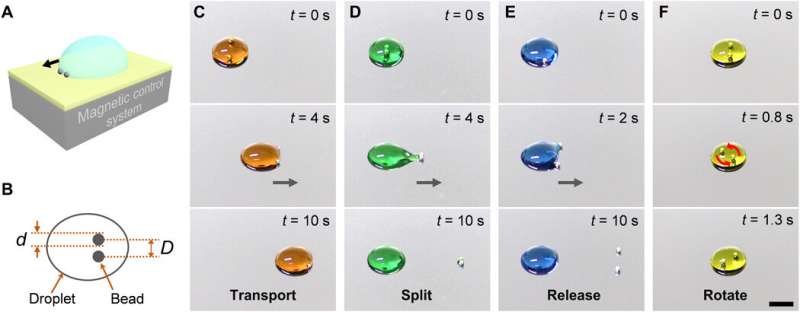
Li et al. named the two steel beads in the magnetic control system the 'robot' and colored the water droplets with food additives to distinctly identify different modes of actuation. The hydrophilic (water-loving) robot easily captured the droplet on contact. The research team transferred the droplet and adjusted its structure to split a daughter drop or release the droplet by reducing or increasing the distance between the beads. The process mainly relied on the robotic structure where its volume (V) influenced the result. Li et al. quantified the structure of the robot using the beads' center-to-center distance to the diameter (D/d). For example, a robot with a D/d ratio of 1.67 could transport a 150 µL droplet of water. However, if the droplet enlarged to 350 µL, it would split. To understand this, they mechanically analyzed the entire system and then determined the driving force behind the device to be adhesion between the bead and the droplet.

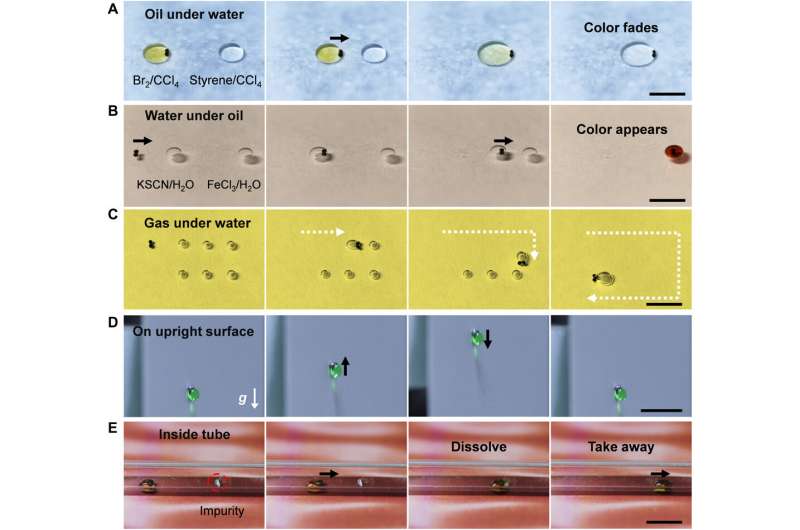
After controlling the water droplet in air, the team investigated the robot under different conditions including diverse atmospheres. For example, they dragged a droplet to move it up or down by overcoming forces of gravity and forces of adhesion between different droplets and the substrate. Such devices have applications in limited spaces such as boxes and tubes vital in microfluidics and clinical medicine. Lossless transport and precise reagent control are vital for quantitative chemical micro-reactions widely employed in analytical chemistry, diagnostics and biotechnology. For example, Li et al. conducted sequential acid-base neutralization reactions via programmable droplet manipulation of the robot, to split a daughter droplet and transfer it to form a neutral droplet. The robots provided a versatile route to automatically control droplets for micro-reactions using precise aliquots of reagent that are typically toxic, radioactive or explosive in nature.
The research scientists then simulated biomedical processes in the lab to explore the potential of the robot during in vivo medical applications. They first simulated calculi or mineral deposits found in organs such as kidney and gallbladder, which generally remain intact due to structural limits that prevent their surgical removal. Using the microrobot, they dragged a drug droplet on to the simulated calculi to the calculi for easy collection and removal (based on the compatibility between the two surfaces). In the next experiment, they showed the robot's potential to remove or clear blood vessels by building up a surfeit of cholesterol in coronary arteries—represented with a tube filled with water. They transported the drug droplets to capture the simulated plaque and dissolved the drug for its disposal by the robot. The innovative methods can have versatile functions for in vivo medical applications.

In this way, An Li and colleagues proposed a simple and general strategy to manipulate droplets using a magnetic-actuated robot made of two steel beads. They controlled the structure using a magnetic field that regulated the distribution of resistive forces at the front and rear segments of the droplet. They realized multiple behaviors for the droplets including transport, split, release and rotation. In addition to manipulating water droplets in air, the robot showed promise to transport complex liquids such as oil, water-in-oil and gas-in-water. The steel beads showed maneuverability of the droplets in limited spaces, including toxic and radioactive environments. The work has significant potential in the fields of device fabrication, sensing and bioassay, as well as in medicine in vivo. The team envision further optimizations of the system will allow smaller droplet manipulation at the nano- and pico-liter scale with enhanced biocompatibility.
More information: An Li et al. Programmable droplet manipulation by a magnetic-actuated robot, Science Advances (2020). DOI: 10.1126/sciadv.aay5808
M. Prakash et al. Microfluidic Bubble Logic, Science (2007). DOI: 10.1126/science.1136907
Nan Gao et al. How drops start sliding over solid surfaces, Nature Physics (2017). DOI: 10.1038/nphys4305
Journal information: Science Advances , Science , Nature Physics
© 2020 Science X Network