Gold's wobbly nucleus: What the short-lived Au187 isotope teaches us about fundamental science research

As Earth rotates along its axis, it wobbles a little bit. This wobbling comes, in part, from how mass is distributed across the planet. Nuclear physics researchers have now observed this same type of wobbling in Au187—a gold isotope that lives for just eight minutes. Fundamental science research like this can lead to major breakthroughs in a range of fields, including medical care.
Robert Janssens, Edward G. Bilpuch Professor of Physics in UNC-Chapel Hill's College of Arts & Sciences, is a member of the research team that observed the wobble. The team published a paper on its findings in Physical Review Letters.
UNC News spoke to Janssens about the findings and why research into fundamental science is so important.
Question: How did you find out that gold 187's nucleus is wobbly?
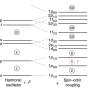
Answer: This gold is not a stable gold. You'd have to really hurry to find it in nature, as it lives a short amount of time—just eight minutes. We create it in the lab through a nuclear reaction. Studying this particular gold isotope we found that it is triaxial, meaning is has three axes of different length, and there are not that many nuclei that are triaxial.
Finding out that the nucleus is triaxial is not easy. One of the best ways to study that is to force the nucleus to spin. When we did that, we found details in the data that told us its shape. And with that, we could demonstrate that in the same way Earth wobbles around one of its axes, this gold wobbles around its longest axis.
Q: What makes it wobbly?
A: Gold 187 has an odd number of nucleons and that's important. Something we've known for a long time about nuclei is that protons and neutrons like to go two-by-two: we call this phenomenon pairing. Except in this case, there's an odd nucleon sitting out there all alone and it's trying to follow the motion of the rotation of all of the others. We think that's what results in the wobbling.
Q: Why is it important to keep exploring basic science topics like this one?
A: We really try to understand the basic properties of the nucleus, like for example its shape. In the Triangle Universities Nuclear Laboratory, we have found nuclei that can adopt three different shapes. At rest or in a ground state, they are spherical. Pump up the energy and they can become oblate, or like the shape of Earth, round but a little compressed. Add more energy and you get an American football shape—we call that prolate.
We'd like to know if nuclei are as universal as we'd like to think, and studying these basics shows us nuclear forces we don't understand or that we haven't seen before. Nuclear physics has so many applications. One being that about 50% of medical diagnostics and therapies are done with nuclear techniques or with radioactive isotopes that were originally discovered by scientists studying them to understand their fundamental properties. While a big part of my job is to do the basic research, I'm very glad that it is so relevant to society.
More information: N. Sensharma et al. Longitudinal Wobbling Motion in Au187, Physical Review Letters (2020). DOI: 10.1103/PhysRevLett.124.052501
Journal information: Physical Review Letters
Provided by University of North Carolina at Chapel Hill