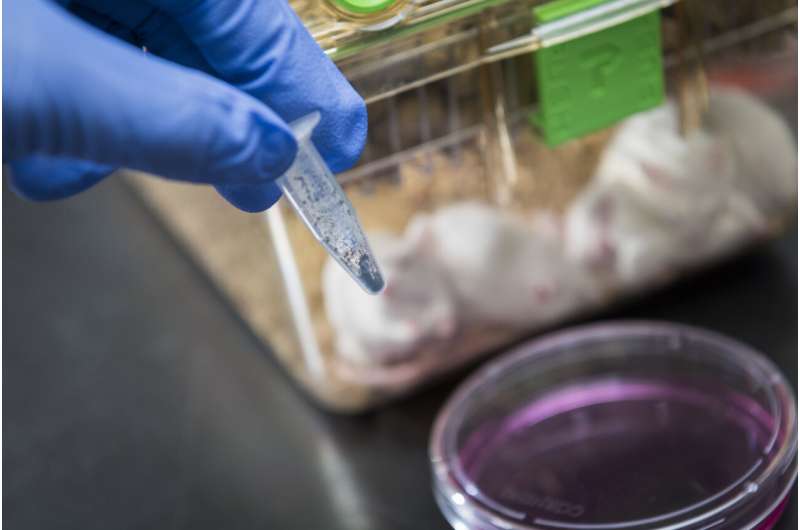
The controllable activation property and superior antitumor activity of phorbiplatin significantly contributes to the development of photoactivatable anticancer prodrugs, especially Pt(IV) prodrugs that can be activated by red light, to reduce the adverse effects and conquer drug resistance of traditional platinum chemotherapy. Credit: Wang et al., Phorbiplatin, a Highly Potent Pt(IV) Antitumor Prodrug That Can Be Controllably Activated by RedLight, Chem (2019), 10.1016/j.chempr.2019.08.021
Most of the current clinical anti-tumor drugs used in chemotherapy move around in the patient's blood after intake and are unable to pinpoint the targeted tumor. As a result, while killing the tumor cells, the healthy cells may also be killed as "collateral damage," leading to undesired side effects. Aiming to overcome this problem, Dr. Zhu Guangyu, Associate Professor of Department of Chemistry, City University of Hong Kong (CityU) and his research team have recently developed phorbiplatin, an anti-cancer prodrug that can be controllably activated by red light. With its unique "on-site" activation characteristic, it will effectively kill cancer cells and minimize damage to normal tissues.
Phorbiplatin is a small-molecule platinum (IV) anti-cancer prodrug. Prodrug is a compound that will only be pharmacologically active after activation inside the body. Phorbiplatin is shown to be inert in the dark but can be activated under low-power red-light irradiation. "Under short-period irradiation with low intensity of red light (650 nm, 7 mW/cm2) and without any external catalyst, phorbiplatin is reduced to oxaliplatin, a first-line clinical chemotherapeutic drug, as well as pyropheophorbide a (PPA), a photoactivation ligand. Both substances are effective in killing tumor cells," Dr. Zhu explained the controllable activation property.
"Phorbiplatin is the first small-molecule platinum (IV) prodrug that can be activated by a red light," he added.
Unique photoreduction mechanism first discovered
Previously, researchers utilized ultraviolet light to activate small-molecule platinum anti-tumor drugs. However, ultraviolet light has poor penetration depth and can also damage cells. Therefore related experiments have not yet been conducted in vivo. On the contrary, "red light does not harm normal cells. And it possesses higher penetration depth through the skin to reach the subcutaneous tumor," Dr. Zhu explained, regarding his choice of red light in this research.
It took the team around 3 years to discover and study phorbiplatin, which is scaffolded on the molecular structure of oxaliplatin, a first-line anti-tumor drug widely used in the treatment of different cancers, such as colorectal cancer, gastric cancer, and ovarian cancer. The team functionalized oxaliplatin with PPA, which is a photosensitizer in photodynamic therapy and highly sensitive to red light, as the photoactive ligand to obtain phorbiplatin. Under irradiation with low intensity of red light, PPA acts as a "photo-induced redox relay" to transfer electrons from reducing agents to the platinum (IV) center to facilitate the reduction process, and as a result, releasing oxaliplatin.
Citing the statistics from the research, Dr. Zhu said only a short period of irradiation would trigger the photoreduction mechanism. In just 10 minutes, 81% of phorbiplatin was reduced rapidly to oxaliplatin and PPA. Phorbiplatin is controllably activated by red light in a spatial and temporal fashion and holds promise for minimizing the side effects originating from non-specific activation.
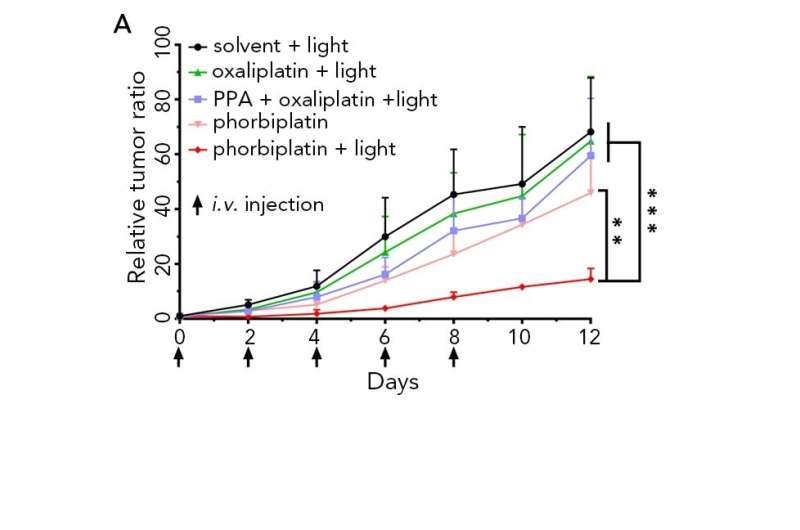
Phorbiplatin (red line) displays remarkable ability to kill breast cancer cells in mice, results showed much higher photocytotoxicity compared with oxaliplatin (green line). Credit: Wang et al., Phorbiplatin, a Highly Potent Pt(IV) Antitumor Prodrug That Can Be Controllably Activated by RedLight, Chem (2019), 10.1016/j.chempr.2019.08.021
Significantly improved anti-tumor activity
The team also examined the cytotoxicity of phorbiplatin to different tumor cells. They found that platinum-sensitive (A2780) human ovarian cancer cells treated with phorbiplatin under red light irradiation showed high fractions of dead (68.0%) cells. Compared with oxaliplatin, phorbiplatin displayed a remarkable ability to kill human breast cancer cells (MCF-7) with up to a 1,786-fold increase in photocytotoxicity, and a 974-fold increase for platinum-resistant (A2780cisR) human ovarian cancer cells.
Moreover, phorbiplatin with irradiation significantly inhibited tumor growth in mice, with 67% reduction in tumor volume and 62% reduction in tumor weight compared with mice treated with oxaliplatin, PPA, and even a mixture of oxaliplatin and PPA. The body weight of the mice treated with phorbiplatin did not change significantly, highlighting its safety.
Dr. Zhu stressed that phorbiplatin is kinetically stable in the dark. In their toxicity tests, it indicated low toxicity to the normal human lung fibroblasts (MRC-5) in the dark. And for the mice treated with phorbiplatin under irradiation, their organs like heart, liver, spleen, lung and kidney were in good condition, further confirming the safety of phorbiplatin, while there was liver damage in the mice treated directly with the mixture of oxaliplatin and PPA under irradiation.
A window for development of photoactivatable prodrugs
Dr. Zhu suggested that the controllable activation property and superior anti-tumor activity of phorbiplatin significantly contribute to the development of photoactivatable anti-cancer prodrugs, especially platinum (IV) prodrugs that can be activated by red light, to reduce the adverse effects and conquer drug resistance of traditional platinum chemotherapy. The team will work on pre-clinical study and conduct more toxicity tests as well as efficacy tests. He believed that due to the improved molecular structure, "it is easier for phobriplatin to enter the tumor and accumulate, causing DNA damage and eventually kill the tumor," in a way to overcome drug resistance issues.
More information: Zhigang Wang et al, Phorbiplatin, a Highly Potent Pt(IV) Antitumor Prodrug That Can Be Controllably Activated by Red Light, Chem (2019). DOI: 10.1016/j.chempr.2019.08.021
Journal information: Chem
Provided by City University of Hong Kong