Evolving alongside other bacteria keeps hospital bug potent

Bacteria that evolve in natural environments—rather than laboratory tests—may become resistant to phage treatments without losing their virulence, new research shows.
The University of Exeter study examined why Pseudomonas aeruginosa bacteria develop resistance to phages (viruses that kill bacteria) in different ways in labs and the real world.In labs, bacterial evolution tends to result in the loss of a receptor to which phages usually attach. In natural environments, the bacteria tend to evolve resistance based on an immune mechanism called CRISPR-Cas.
The tests were conducted in a lab, but by introducing other bacteria alongside Pseudomonas aeruginosa—a common cause of infections in hospitals—the researchers showed that additional "biotic complexity" tipped the balance in favour of CRISPR-based resistance.
And they found that—unlike evolving to lose phage receptors—this CRISPR-based resistance did not reduce the bacteria's virulence (the severity of effects).
"When we introduced biodiversity, the bacteria shifted towards favouring the CRISPR-based evolution," said lead author Ellinor Alseth, of the Environment and Sustainability Institute on Exeter's Penryn Campus in Cornwall.
"In more natural environments, losing the surface receptor (to which phages attach) comes with a cost because it serves other functions.
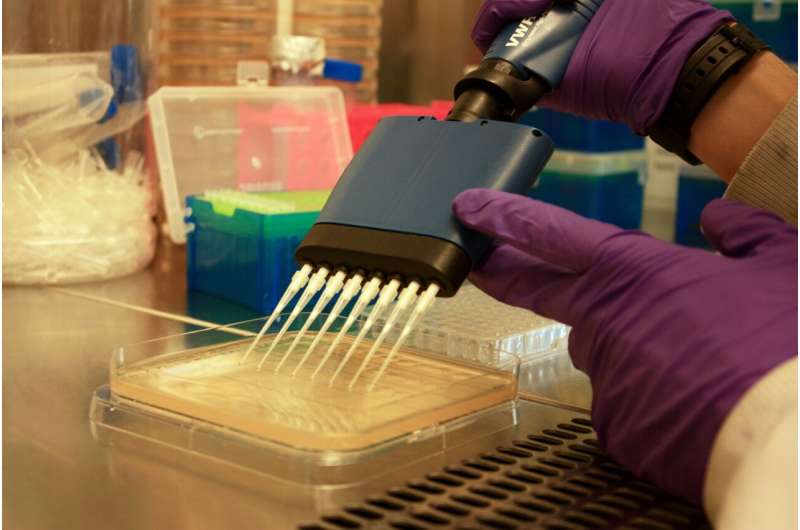
"Without this surface receptor, the bacteria suffer a 'fitness cost' and are less competitive compared to other kinds of bacteria.
"So, in a more complex—more 'natural' - environment, CRISPR-based resistance is favoured.
"With the CRISPR evolutionary route, the bacteria maintain their level of virulence rather than becoming less virulent."Virulence was tested by observing the effects of the bacteria on moth larvae (Galleria mellonella), but the researchers say similar effects would be expected in humans. Pseudomonas aeruginosa thrives in a range of different environments, including hospitals. It frequently colonises the lungs of cystic fibrosis patients, in whom it is the leading cause of morbidity and death.
The Exeter team studied the effects of a particular phage (DMS3vir) which is known to kill this bacteria.
The findings demonstrate that the evolutionary outcome of bacteria-phage interactions can be fundamentally altered by the microbial community context.
Such interactions are traditionally studied in isolation, but the researchers say it is "increasingly clear" that the presence of a complex mix of other microbes can have "key implications for the evolutionary epidemiology of infectious disease".
The paper, published in the journal Nature, is entitled: "Bacterial biodiversity drives the evolution of CRISPR-based phage resistance."
More information: Bacterial biodiversity drives the evolution of CRISPR-based phage resistance, Nature (2019). DOI: 10.1038/s41586-019-1662-9 , nature.com/articles/s41586-019-1662-9
Journal information: Nature
Provided by University of Exeter