Imaging the chemical structure of individual molecules, atom by atom

For physicist Percy Zahl, optimizing and preparing a noncontact atomic force microscope (nc-AFM) to directly visualize the chemical structure of a single molecule is a bit like playing a virtual reality video game. The process requires navigating and manipulating the tip of the instrument over the world of atoms and molecules, eventually picking some up at the right location and in the right way. If these challenges are completed successfully, you advance to the highest level, obtaining images that precisely show where individual atoms are located and how they are chemically bonded to other atoms. But take one wrong move, and it is game over. Time to start again.
"The nc-AFM has a very sensitive single-molecule tip that scans over a carefully prepared clean single-crystal surface at a constant height and "feels" the forces between the tip molecule and single atoms and bonds of molecules placed on this clean surface," explained Zahl, who is part of the Interface Science and Catalysis Group at the Center for Functional Nanomaterials (CFN), a U.S. Department of Energy (DOE) Office of Science User Facility at Brookhaven National Laboratory. "It can take an hour or days to get this sensor working properly. You can't simply press a button; fine tuning is required. But all of this effort is definitely worthwhile once you see the images appearing like molecules in a chemistry textbook."
A history of chemical structure determination
Since the beginning of the field of chemistry, scientists have been able to determine the elemental composition of molecules. What has been more difficult is to figure out their chemical structures, or the particular arrangement of atoms in space. Knowing the chemical structure is important because it impacts the molecule's reactivities and other properties.
For example, Michael Faraday isolated benzene in 1825 from an oil gas residue. It was soon determined that benzene is composed of six hydrogen and six carbon atoms, but its chemical structure remained controversial until 1865, when Friedrich August Kekulé proposed a cyclic structure. However, his proposal was not based on a direct observation but rather on logic deduction from the number of isomers (compounds with the same chemical formula but different chemical structures) of benzene. The correct symmetric hexagonal structure of benzene was finally revealed through its diffraction pattern obtained by Kathleen Lonsdale via X-ray crystallography in 1929. In 1931, Erich Huckel used quantum theory to explain the origin of "aromaticity" in benzene. Aromaticity is a property of flat ring-shaped molecules in which electrons are shared between atoms. Because of this unique arrangement of electrons, aromatic compounds have a special stability (low reactivity).
Today, X-ray crystallography continues to be a mainstream technique for determining chemical structures, along with nuclear magnetic resonance spectroscopy. However, both techniques require crystals or relatively pure samples, and chemical structure models must be deducted by analyzing the resulting diffraction patterns or spectra.
The first-ever actual image of a chemical structure was obtained only a decade ago. In 2009, scientists at IBM Research–Zurich Lab in Switzerland used nc-AFM to resolve the atomic backbone of an individual molecule of pentacene, seeing its five fused benzene rings and even the carbon-hydrogen bonds. This breakthrough was made possible by selecting an appropriate molecule for the end of the tip—one that could come very close to the surface of pentacene without reacting with or binding to it. It also required optimized sensor readout electronics at cryogenic temperatures to measure small frequency shifts in the probe oscillation (which relates to the force) while maintaining mechanical and thermal stability through vibration damping setups, ultrahigh vacuum chambers, and low-temperature cooling systems.
"Low-temperature nc-AFM is the only method that can directly image the chemical structure of a single molecule," said Zahl. "With nc-AFM, you can visualize the positions of individual atoms and the arrangement of chemical bonds, which affect the molecule's reactivity."
However, currently there are still some requirements for molecules to be suitable for nc-AFM imaging. Molecules must be mainly planar (flat), as the scanning occurs on the surface and thus is not suitable for large three-dimensional (3-D) structures such as proteins. In addition, because of the slow nature of scanning, only a few hundred molecules can be practically examined per experiment. Zahl notes that this limitation could be overcome in the future through artificial intelligence, which would pave the way toward automated scanning probe microscopy.
According to Zahl, though nc-AFM has since been applied by a few groups around the world, it is not widespread, especially in the United States.
"The technique is still relatively new and there is a long learning curve in acquiring CO tip-based molecular structures," said Zahl. "It takes a lot of experience in scanning probe microscopy, as well as patience."
A unique capability and expertise
The nc-AFM at the CFN represents one of a few in this country. Over the past several years, Zahl has upgraded and customized the instrument, most notably with the open-source software and hardware, GXSM (for Gnome X Scanning Microscopy). Zahl has been developing GXSM for more than two decades. A real-time signal processing control system and software continuously records operating conditions and automatically adjusts the tip position as necessary to avoid unwanted collisions when the instrument is operated in an AFM-specific scanning mode to record forces over molecules. Because Zahl wrote the software himself, he can program and implement new imaging or operating modes for novel measurements and add features to help operators better explore the atomic world.
For example, recently Zahl applied a custom "slicing" mode to determine the 3-D geometrical configuration in which a single molecule of dibenzothiopene (DBT)—a sulfur-containing aromatic molecule commonly found in petroleum—adsorbs on a gold surface. The DBT molecule is not entirely planar but rather tilted at an angle, so he combined a series of force images (slices) to create a topographic-like representation of the molecule's entire structure.
"In this mode, obstacles such as protruding atoms are automatically avoided," said Zahl. "This capability is important, as the force measurements are ideally taken in one fixed plane, with the need to be very close to the atoms to feel the repulsive forces and ultimately to achieve detailed image contrast. When parts stick out of the molecule plane, they will likely negatively impact image quality."
This imaging of DBT was part of a collaboration with Yunlong Zhang, a physical organic chemist at ExxonMobil Research and Engineering Corporate Strategic Research in New Jersey. Zhang met Zahl at a conference two years ago and realized that the capabilities and expertise in nc-AFM at the CFN would have great potential for his research on petroleum chemistry.
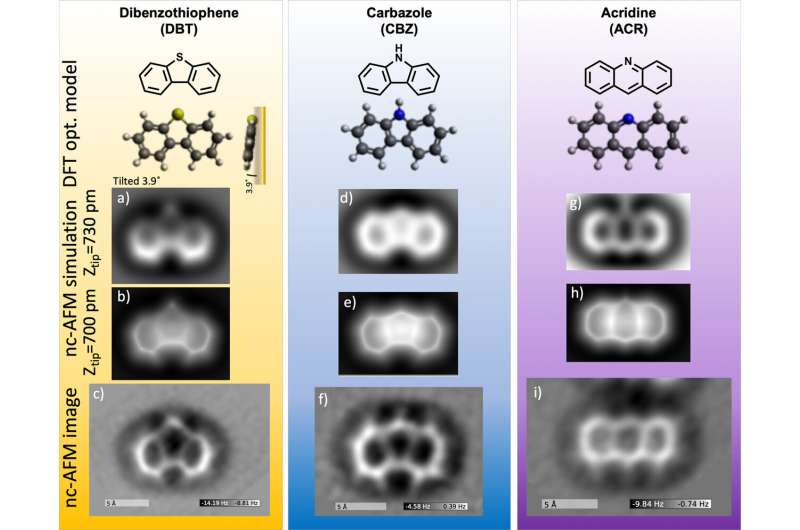
Zahl and Zhang used nc-AFM to image the chemical structure of not only DBT but also of two nitrogen-containing aromatic molecules—carbazole (CBZ) and acridine (ACR)—that are widely observed in petroleum. In analyzing the images, they developed a set of templates of common features in the ring-shaped molecules that can be used to find sulfur and nitrogen atoms and distinguish them from carbon atoms.
Petroleum: a complex mixture
The chemical composition of petroleum widely varies depending on where and how it formed, but in general it contains mostly carbon and hydrogen (hydrocarbons) and smaller amounts of other elements, including sulfur and nitrogen. During combustion, when the fuel is burned, these "heteroatoms" produce sulfur and nitrogen oxides, which contribute to the formation of acid rain and smog, both air pollutants that are harmful to human health and the environment. Heteroatoms can also reduce fuel stability and corrode engine components. Though refining processes exist, not all of the sulfur and nitrogen is removed. Identifying the most common structures of impure molecules containing nitrogen and sulfur atoms could lead to optimized refining processes for producing cleaner and more efficient fuels.
"Our previous research with the IBM group at Zurich on petroleum asphaltenes and heavy oil mixtures provided the first "peek" into numerous structures in petroleum," said Zhang. "However, more systemic studies are needed, especially on the presence of heteroatoms and their precise locations within aromatic hydrocarbon frameworks in order to broaden the application of this new technique to identify complex molecular structures in petroleum."
To image the atoms and bonds in DBT, CBZ, and ACR, the scientists prepared the tip of the nc-AFM with a single crystal of gold at the apex and a single molecule of carbon monoxide (CO) at the termination point (the same kind of molecule used in the original IBM experiment). The metal crystal provides an atomically clean and flat support from which the CO molecule can be picked up.
After "functionalizing" the tip, they deposited a few of each of the molecules (dusting amount) on a gold surface inside the nc-AFM under ultrahigh vacuum at room temperature via sublimation. During sublimation, the molecules go directly from a solid to gas phase.
Though the images they obtained strikingly resemble chemical structure drawings, you cannot directly tell from these images whether there is a nitrogen, sulfur, or carbon atom present in a particular site. It takes some input knowledge to deduct this information.
"As a starting point, we imaged small well-known molecules with typical building blocks that are found in larger polycyclic aromatic hydrocarbons—in this case, in petroleum," explained Zahl. "Our idea was to see what the basic building blocks of these chemical structures look like and use them to create a set of templates for finding them in larger unknown molecular mixtures."

For example, for sulfur- and nitrogen-containing molecules in petroleum, sulfur is only found in ring structures with five atoms (pentagon ring structure), while nitrogen can be present in rings with either five or six (hexagonal ring structure) atoms. In addition to this bonding geometry, the relative "size," or atomic radius, of the elements can help distinguish them. Sulfur is relatively larger than nitrogen and carbon, and nitrogen is slightly smaller than carbon. It is this size, or "height," that AFM is extremely sensitive to.
"Simply speaking, the force that the AFM records in very close proximity to an atom relates to the distance and thus to the size of that atom; as the AFM scans over a molecule at a fixed elevation, bigger atoms protrude more out of the plane," explained Zahl. "Therefore, the larger the atom in a molecule, the bigger the force that the AFM records as it gets closer to its atomic shell, and the repulsion increases dramatically. That is why in the images sulfur appears as a bright dot, while nitrogen looks a hint fainter."
Zahl and Zhang then compared their experimental images to computer-simulated ones they obtained using the mechanical probe particle simulation method. This method simulates the actual forces acting on the CO molecule on the tip end as it scans over molecules and bends in response. They also performed theoretical calculations to determine how the electrostatic potential (charge distribution) of the molecules affects the measured force and relates to their appearance in the nc-AFM images.
"We used density functional theory to study how the forces felt by the CO probe molecule behave in the presence of the charge environment surrounding the molecules," said Zahl. "We need to know how the electrons are distributed in order to understand the atomic force and bond contrast mechanism. These insights even allow us to assign single or double bonds between atoms by analyzing image details."
Going forward, Zahl will continue developing and enhancing nc-AFM imaging modes and related technologies to explore many kinds of interesting, unknown, or novel molecules in collaboration with various users. Top candidate molecules of interest include those with large magnetic moments and special spin properties for quantum applications and novel graphene-like (graphene is a one-atom-thick sheet of carbon atoms arranged in a hexagonal lattice) materials with extraordinary electronic properties.
"The CFN has unique capabilities and expertise in nc-AFM that can be applied to a wide range of molecules," said Zahl. "In the coming years, I believe that artificial intelligence will make a big impact on the field by helping us operate the microscope autonomously to perform the most time-consuming, tedious, and error-prone parts of experiments. With this special power, our chances of winning the "game" will be much improved."
More information: Percy Zahl et al. Guide for Atomic Force Microscopy Image Analysis To Discriminate Heteroatoms in Aromatic Molecules, Energy & Fuels (2019). DOI: 10.1021/acs.energyfuels.9b00165
Provided by Brookhaven National Laboratory