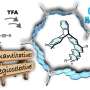
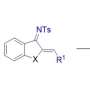
We developed an efficient method for the diastereoselective synthesis of CF3-substituted spiroisochromans via C(sp3)¬-H bond functionalization involving sequential transformations ([1,5]-hydride shift/cyclization/elimination of MeOH/intramolecular Friedel-Crafts reaction). The salient feature of this reaction is that diastereoselectivity was affected by the subtle structural difference of the starting materials. This method gave the stereochemically defined CF3-substituted spiroisochromans, which is otherwise difficult to synthesize by the conventional method, and highlight the high synthetic potential of the internal redox reaction. Credit: Figure adapted with permission from Org. Lett. 2019, 21, 2383?2387. © 2019 AMERICAN CHEMICAL SOCIETY
Japanese scientists have—for the first time—developed an efficient way to make organic molecules that have so far been difficult to synthesize because of their overall bulky structure and general instability.
By designing a carefully planned sequence of synthesis steps, the researchers were able to efficiently create intricate molecules that have several practical properties. Potential applications include tool compounds for biomedical study and to serve as scaffolds for the design of therapeutic agents with new shapes to materials designs, specifically in the creation of dyes and electronically active materials
The research findings were published in Organic Letters in March 2019.
Designing complex organic molecules that can be of use in a wide range of applications is not always straightforward. Often, these molecules are complex, meaning that they are a combination of several cyclical structures that are all attached to one atom. While beautiful, their synthesis is difficult for several reasons. Their bulkiness makes them energetically unfavorable—or unstable—which means they are difficult to synthesize.
On the other hand, if they are eventually synthesized, there is not enough compound to be of use for any subsequent application. An example of such complex molecules are those that involve several cyclic structures that are attached to carbon-hydrogen atoms within it. Previous methods have relied not only on several reaction steps but have resulted in very little compound yield.
Now, Keiji Mori, Ph.D., Associate Professor at the Department of Applied Chemistry, Graduate School of Engineering, Tokyo University of Agriculture and Technology, Japan and his team have come up with a straightforward method that overcomes the shortcomings of previous synthesis sequences.
The researchers focused on the carbon-hydrogen bond within their starting step and proceeded with a series of bond editions that involved sequential transformations of the molecule. After achieving the desired cyclic conformations, the scientists introduced a few more carbon-hydrogen groups to obtain an ultimate molecule with the energetically preferred conformation. "It is important to note that even the simplest idea can lead to an important synthetic method. Along those lines, subtle differences in the substrate can dramatically alter the reaction mechanism," adds Mori.
The researchers hope that this new synthesis will provide access to a host of useful molecules in future. "The synthesis of compounds whose molecular structure is composed of several kinds of circular structures has been difficult to achieve via conventional methods. As such, the final goal of the project is the synthesis of various spiro (or twisted-shaped) compounds that have a variety of different atoms," Mori adds. These new molecules could greatly enhance both medical as well as materials sciences.
This work was partially supported by a Grant-in-Aid for Scientific Research from the Japan Society for the Promotion of Science, and by grants from The Uehara Memorial Foundation and The Naito Foundation.
More information: Risa Tamura et al, Diastereoselective Synthesis of CF3-Substituted Spiroisochromans by [1,5]-Hydride Shift/Cyclization/Intramolecular Friedel–Crafts Reaction Sequence, Organic Letters (2019). DOI: 10.1021/acs.orglett.9b00668
Journal information: Organic Letters
Provided by Tokyo University of Agriculture and Technology
![We developed an efficient method for the diastereoselective synthesis of CF3-substituted spiroisochromans via C(sp3)¬-H bond functionalization involving sequential transformations ([1,5]-hydride shift/cyclization/elimination of MeOH/intramolecular Friedel-Crafts reaction). The salient feature of this reaction is that diastereoselectivity was affected by the subtle structural difference of the starting materials. This method gave the stereochemically defined CF3-substituted spiroisochromans, which is otherwise difficult to synthesize by the conventional method, and highlight the high synthetic potential of the internal redox reaction. Credit: Figure adapted with permission from Org. Lett. 2019, 21, 2383?2387. © 2019 AMERICAN CHEMICAL SOCIETY New synthesis of complex organic molecules revealed](https://scx1.b-cdn.net/csz/news/800a/2019/2-newsynthesis.jpg)