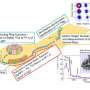
Nuclear 'magic numbers' collapse beyond the doubly magic nickel 78

Scientists from the RIKEN Nishina Center for Accelerator-Based Research and collaborators have used the center's heavy ion accelerator, the RI Beam Factory, to demonstrate that nickel-78, a neutron-rich "doubly magic" isotope of nickel with 28 protons and 50 neutrons, still maintains a spherical shape that makes it relatively stable despite the large imbalance in the number of protons and neutrons. They also discovered a surprise—observations from the experiment suggest that nickel-78 may be the lightest nucleus with 50 neutrons to have a magic nature. Lighter isotones—meaning nuclei with the same number of neutrons but different number of protons—would inevitably be deformed, despite having the magic number of neutrons.
Grasping the validity of the magic numbers in extremely neutron-rich nuclei is crucial to understanding why our universe has the mix of nuclei that we see today. Elements heavier than iron are not synthesized in the normal burning of stars, but are mainly created through two processes known as the s-process and r-process, which involve nuclei capturing extra neutrons. The r-process, in which neutrons are rapidly absorbed, is particularly important as it is responsible for the creation of certain neutron-rich nuclei. During the process, nuclei accumulate neutrons until they reach a state in which they can no longer accept them—this state is known as a waiting point—and then undergo a process known as beta decay, in which they lose a neutron but gain a proton, allowing them to begin to accept new neutrons. The r-process, which accounts for about half of the production of nuclei heavier than iron, can only take place in extraordinary neutron-rich environments such as supernova explosions and neutron star mergers like the one that was observed in 2017.
The precise location of these "waiting points" is not well understood, however. Complicating the process is that magic numbers of either protons or neutrons—equivalent to the idea of closed electron shells in chemistry—make the nuclei more resistant to capturing further neutrons. One well-known magic number is 50 neutrons, but it has been unclear whether this number is preserved for extremely neutron-rich nuclei.
To get an answer, the group decided to experiment with nickel-78, a doubly magic isotope that has only recently become accessible to experimentation thanks to powerful accelerators such as the RI Beam Factory in Japan, the one used in this study. To perform the experiment, published in Nature, the researchers combined observations from the MINOS detector operated by CEA in France and the DALI2 detector operated by RIKEN, both located within the RIBF complex. They generated a beam of uranium-238 and used it to bombard a target of beryllium, forcing the uranium to fission into isotopes such as copper-79 and zinc-80—both of which have 50 neutrons.
These two beams were then sent to hit a hydrogen target, sometimes producing nickel-78, the focus of the research.
Using gamma ray detectors, the group demonstrated that nickel-78 is relatively stable, as predicted by calculations, maintaining a spherical rather than deformed shape. Ryo Taniuchi of the University of Tokyo and the RIKEN Nishina Center for Accelerator-Based Science says, "We were happy to be able to show experimentally that nickel-78 does maintain the spherical shape that calculations predicted it would. We were surprised, however, to discover that the nucleus also has a competing shape, which is not spherical, and that any lighter isotone than the one we used would be subject to this deformation and would not maintain its magic nature."
Pieter Doornenbal of the Nishina Center says, "This is an important finding, as it gives us new insights for how magic numbers appear and disappear across the nuclear landscape and affect the process of nucleosynthesis that led to the abundance of isotopes that we see in the universe today. We intend to do further experiments with even lighter isotones with 50 neutrons to experimentally demonstrate this finding."
More information: 78Ni revealed as a doubly magic stronghold against nuclear deformation, Nature (2019). DOI: 10.1038/s41586-019-1155-x , www.nature.com/articles/s41586-019-1155-x
Journal information: Nature
Provided by RIKEN