Biochemists untangle mysteries of cellular form, function

The complex inner workings of cells, from their architecture to their signaling, underlie much of multicellular organic life. How are they built? How do their proteins interact? And most crucially, how can understanding these functions improve our knowledge of biological outcomes such as disease?
University of Colorado Boulder Distinguished Professors Karolin Luger and Natalie Ahn have studied questions such as these for decades. Last year, both were elected to the National Academy of Sciences, one of the most prestigious honors a scientist can receive. The duo will be formally inducted on Saturday, April 27 at the organization's annual meeting.
"It's a high honor because it comes from peers," said Luger, the endowed chair of CU Boulder's Department of Biochemistry and a Howard Hughes Medical Institute Investigator. "It's primarily a wonderful acknowledgement of the collective work of all the former and present students, post-docs and technicians who have contributed to this research."
Like an archeologist piecing together the origins of ancient structures, Luger and her students examine the fundamental building blocks of genomic processes and untangle their cellular machinery.
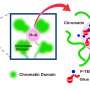
Luger began her career with an interest in X-ray crystallography, a technique used to discern 3-D molecular structures. Eventually, her focus shifted to chromatin, the material that holds DNA, RNA and proteins together in a compact package within eukaryotic cells. As recently as the late 1980s, before the advent of the Human Genome Project, chromatin was thought to be unimportant, similar to packaging material that only serves to hold more valuable items inside.
"It was a binary mentality back then, but it turned out to be much messier, with lots of variation between individual cells," Luger said. "The packaging, so to speak, has very important implications for how cell types differentiate."
Imagine a space filled with labeled cardboard boxes full of books, she says. By reading the labels on the boxes, humans can discern which boxes they'll need soon and which ones they can safely stash away. Chromatin operates similarly: A fertilized egg cell needs everything—all the genomic information it can get—whereas a more mature cell, such as a liver cell, can read the packaging and know what it can safely ignore.
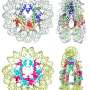
Early advances in electron microscopy revealed chromatin's elegant structure, which appears as "beads on a string," dotted with nucleosomes. Luger went on to determine the structure of nucleosomes at near-atomic resolution, revealing the structure of DNA in all multicellular organisms
More recently, Luger has been examining how and why many multicellular organisms—humans, yeast, trees—all fold their DNA using the same molecular mechanisms. In 2017, her lab and collaborators identified the microbes Archaea (which predate multicellular organisms by 3 billion years) as the likely 'inventors' of genome folding and nucleosome structure that we still observe today.
"I'm always interested in how these structures came about," she said of the evolutionary discovery. "It's a lot of work to bend DNA, and Archaea had developed a nifty system to do this, which was then appropriated and further refined by the first eukaryotic organisms."
Luger credits her students and post-doctoral researchers for their painstaking work on these research topics, adding that she deliberately recruits from different backgrounds such as physics, cell biology and chemistry in order to cultivate interdisciplinary problem-solving.
"Students come in with new ideas for everything and help me view a scientific problem from all angles," she said. "You have to let yourself be challenged."
Ahn arrived at CU Boulder over 25 years ago, bringing expertise in the field known as signal transduction, which involves enzymatic processes that allow cells to respond to external signals. Ahn was the first to describe the enzyme known as mitogen-activated protein kinase kinase (MAP2K), now known to be a crucial activation point in some types of cancer, particularly melanoma. Upon arriving at CU Boulder, she and her new lab proved that aberrant activation of MAP2K causes cancer, making this enzyme a viable target for therapeutic intervention.
The ubiquity of MAP2K in all cells—healthy and diseased alike—led to initial skepticism that it could be a useful drug target, Ahn said, but several cancer-inhibiting treatments focusing on MAP2K have subsequently been approved by the FDA for clinical use.
"Many thought these would never become drug targets because there might be too many side effects," Ahn said. "But it turned out that, remarkably, the drugs are actually very well tolerated, even more so than chemotherapy."
Ahn was also a pioneer in the field of proteomics, which determines the chemistry of proteins by "weighing" molecules using a technology called mass spectrometry. She was an early investigator to adopt proteomics technologies, and use them to study signal transduction. Proteomics is now widely applied in all aspects of biosciences.
For Ahn, her election to the NAS came as a pleasant surprise, the culmination of a long career in basic research that has yielded promising avenues for clinical discoveries.
"I can't quite believe it, but I'm grateful and lucky enough to have really great scientists as colleagues," Ahn said. "The university gave me the space to be creative in my research."
The dual NAS recognition also speaks to the cumulative strength of CU Boulder's biochemistry discipline, which recently became a stand-alone academic department in the College of Arts and Sciences.
"Abraham Lincoln founded the National Academy of Sciences to give trustworthy scientific advice to the President," said Distinguished Professor Thomas Cech, CU Boulder's first Nobel laureate and the director of the BioFrontiers Institute. "Being elected to the NAS is a rare honor, and in a typical year zero or one scientist might be elected from the entire state of Colorado. So for a single department to have two of its faculty elected in the same year is therefore rare and it's worthy of celebration!"
"This is an amazing department with a strong teaching mission," Luger said. "We always have undergraduates participating in the labs who bring a lot of excitement and energy. There is tremendous opportunity here."
Provided by University of Colorado at Boulder