In zebrafish eggs, most rapidly growing cell inhibits neighbors through mechanical signals
"The winner takes it all, the loser standing small"—that's not just true in the famous ABBA song, but also in animal development. Frequently, a group of cells starts out all being the same. But then one cell puts the brake on its neighbors, sending inhibitory signals that stop their differentiation. The "winning" cell, in the end, is different from its neighbors. So far, the only signalling mechanism known to be responsible for such a lateral inhibition was the Notch-Delta signalling pathway. Postdoc Peng Xia and Professor Carl-Philipp Heisenberg at the Institute of Science and Technology Austria (IST Austria), have now described a new mechanism for lateral inhibition in a publication in today's edition of Cell. In zebrafish ovarian follicles, granulosa cells in the envelope that surround the oocyte do not use the Notch-Delta signaling pathway for lateral inhibition, but compete mechanically—the winning cell grows more rapidly and inhibits the growth of its neighbors by mechanically compressing them, thereby becoming the sole micropyle precursor cell—a cell that later plays an important role for the fertilisation of the egg.
Biggest cell compresses its neighbors to inhibit TAZ signalling
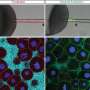
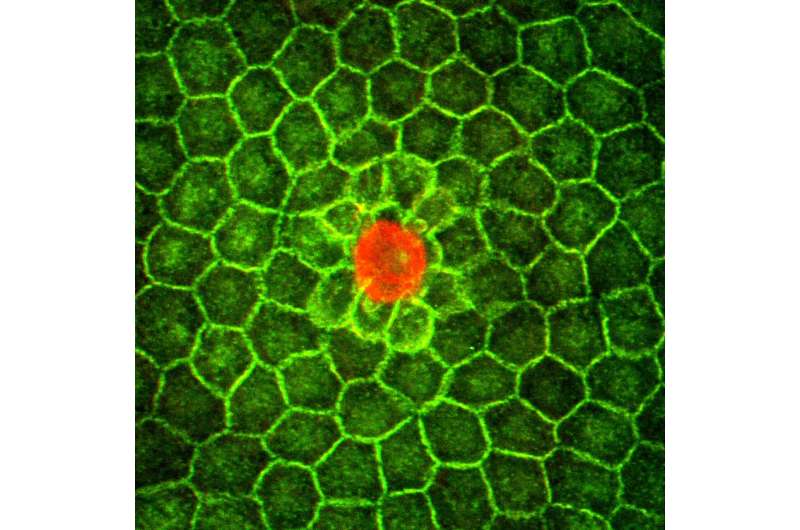
Granulosa cells surround the zebrafish egg. One cell among this group becomes markedly different; it is much bigger than all the others—this is the micropyle precursor cell (MPC). Postdoc Peng Xia and Carl-Philipp Heisenberg set out to understand how the MPC becomes differentiated among all the granulosa cells that could adopt this fate.
Early experiments showed that the Notch-Delta signaling pathway, which is responsible for lateral inhibition in most situations during development, is not involved in the process that specifies the MPC. However, the way in which the MPC grows and presses against its neighboring cells set the researchers on their next path: They decided to test whether this jostling for space plays a role.
With their experimental set-up, the researchers measured tension in the developing tissue, and found that the MPC clearly compresses its neighbors. The researchers then cut out the MPC to observe what happens in its absence.
Normally, as the MPC develops, a signaling pathway that responds to mechanical stimuli called the YAP/TAZ pathway is active in the MPC, while it is less active in the surrounding cells. Once the researchers took out the MPC, the TAZ protein became active again in the surrounding cells. And the cells began to compete with each other again: one cell grew increasingly larger, winning the race to become the new MPC. "This mimics the natural process that we see as the egg develops. When we take out the MPC, another cell will take its role, again by growing rapidly and compressing its neighbors", says first author Peng Xia. "In MPC development, lateral inhibition does not go via the Notch pathway, but is mediated by mechanical signals that control how active the TAZ protein is."

Micropyle forms tunnel through egg shell to allow fertilization
Xia and Heisenberg also uncovered the reason why the MPC becomes so big. TAZ signaling in the MPC not only triggers cell growth but also leads to extracellular matrix deposition around the MPC, which in turn through Integrin adhesion receptors promotes TAZ activity within the MPC. This positive feedback loop eventually leads to TAZ hyperactivation in MPC and its rapid growth.
The large size of the MPC is critical for its future function in forming a tunnel through the egg shell surrounding the oocyte, through which the sperm can reach the oocyte for fertilization. In fish with a mutation in TAZ, the MPC doesn't develop properly. The eggs either have no tunnel or strangely formed tunnels, and the sperm can't get into them—hence the cells are not fertilized, solving the question why this fish line is infertile.
More information: Peng Xia et al, Lateral Inhibition in Cell Specification Mediated by Mechanical Signals Modulating TAZ Activity, Cell (2019). DOI: 10.1016/j.cell.2019.01.019
Journal information: Cell
Provided by Institute of Science and Technology Austria