Tailoring the surface of carbon may hold the key to monitoring patient blood in real-time
The potential applications for tailor-made carbon surfaces are wide and include protective coatings, car parts, biomedical coatings and biosensors. Yet for these developments to be realised, detailed atomic level knowledge is still needed on how carbon surfaces are structured and how they can be modified.
Thanks to the development of a new computational model, Postdoctoral Researcher Miguel Caro is spearheading work in this field by researchers at Aalto University, who work in partnership with Professor Gábor Csányi and Dr. Volker Deringer from Cambridge University.
"For the first time, we can identify the chemical properties of carbon surfaces and better understand how we can prepare them for specific purposes," explains Aalto University's Professor Tomi Laurila.
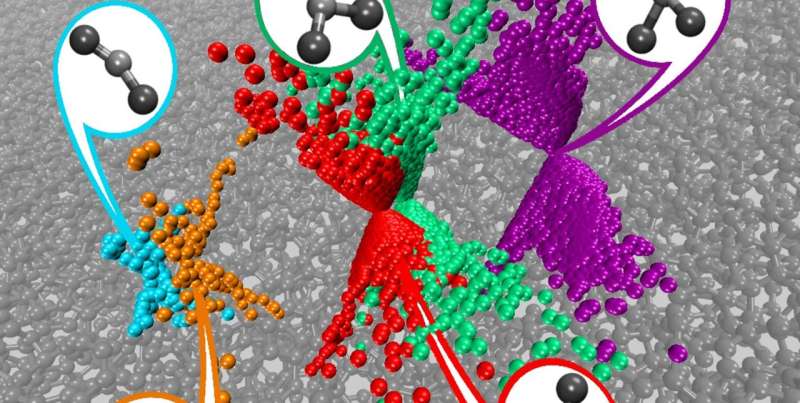
The local environment of every atom in amorphous carbons, also called diamond-like carbons, is slightly different. This means that the number of neighbouring atoms, as well as the distances and angles between them, varies, posing a big challenge in the search to customize these surfaces.
The new computational model has finally allowed researchers to identify a wide variety of local atomic environments and classify them according to their properties. The research team has also calculated the varying degrees of strength with which different groups—hydrogen, alcohol (hydroxyl), and oxygen—will attach to surface sites. Some bonds are, naturally, stronger than others. Because new information about the surface structures can be incorporated to 'retrain' and improve the model, the properties of still unknown surfaces can be predicted based on previous results.
"Through computations, we can now not only explore what material surfaces look like at the atomic level but also see how they interact with other substances under analysis, as well as understand the kinds of chemical groups formed on these surfaces because of this interaction. We are also investigating what kinds of surfaces are needed to optimise the interaction with molecules that we would like to be able to detect, such as hydrogen peroxide," explains Laurila.
In other words, these simulation models based on density functional theory and machine learning tell us what kinds of structures can be developed—and how those structures may be optimised for specific applications.
"In the future we will be able to produce tailored carbon surfaces, for example, for medical sensors, which could be used to monitor the concentration of a particular medication in a patient's blood in real-time. Tracking changes in specific biomarkers in patients may be the key to improving therapeutic treatments currently used, or help us identify the risk of outbreaks of many common diseases earlier than ever before," Laurila says.
The study was published today in Chemistry of Materials.
More information: Miguel A. Caro et al. Reactivity of Amorphous Carbon Surfaces: Rationalizing the Role of Structural Motifs in Functionalization Using Machine Learning, Chemistry of Materials (2018). DOI: 10.1021/acs.chemmater.8b03353
Journal information: Chemistry of Materials
Provided by Aalto University