Scientists discover genetic basis for how harmful algal blooms become toxic

A team led by scientists from Scripps Institution of Oceanography at the University of California San Diego and the J. Craig Venter Institute (JCVI) has uncovered the genetic basis for the production of domoic acid, a potent neurotoxin produced by harmful algal blooms.
Harmful algal blooms cause significant economic and environmental damage to coastal communities around the world. These blooms occasionally produce toxins that can sicken marine mammals and can threaten human health when the toxins accumulate in seafood. A high-dose exposure to domoic acid, produced by a type of phytoplankton known as diatoms in the genus Pseudo-nitzschia, can lead to amnesic shellfish poisoning, a potentially fatal condition characterized by seizures and short-term memory loss.
In a new study appearing in the Sept. 28 edition of Science, the team of UC San Diego and JCVI scientists identified a cluster of genes associated with production of the toxin domoic acid in the marine phytoplankton Pseudo-nitzschia.
This type of microalga is noteworthy because in the summer of 2015 it caused the largest harmful algal bloom ever recorded off the West Coast of North America, from Alaska to Santa Barbara, and resulted in the closure of fisheries and crabbing seasons to protect consumers from potential shellfish poisoning.
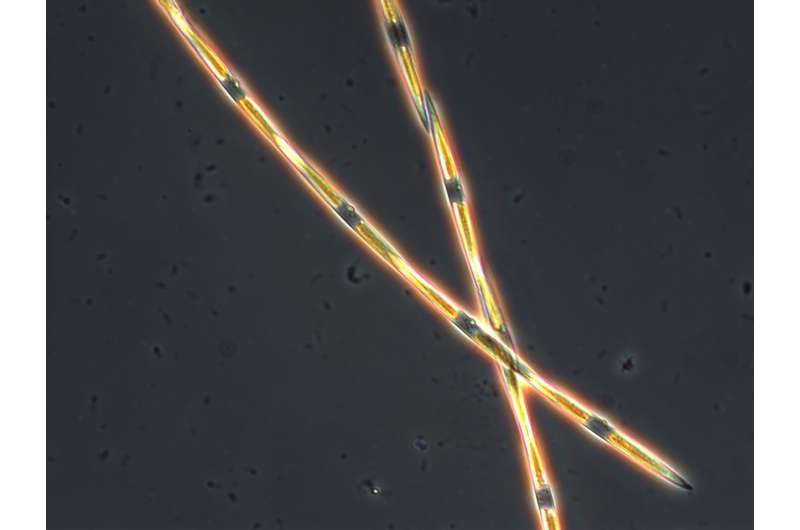
Despite decades of research on Pseudo-nitzschia, the molecular basis for the toxicity of these phytoplankton was not known. Scientists found that these newly discovered genes contain the biological instructions for making the toxin and are subsequently "turned on" when Pseudo-nitzschia is producing domoic acid.
"By identifying the genes that encode domoic acid production, we can now ask questions about various oceanic conditions that turn the genes on or off," said Scripps Institution of Oceanography and JCVI Ph.D. student Patrick Brunson, one of two lead authors on the study. "This knowledge will allow us to track the development of bloom toxicity at the genetic level."
By showing how the genes for domoic acid production are turned on in culture, the authors suggest a way to connect the oceanic conditions that drive algal bloom evolution to the development of toxin production.
"Understanding how algae blooms become toxic and what conditions cause that is critically important," says Hedy Edmonds, a program director in the National Science Foundation's Division of Ocean Sciences, which partially funded the research. "This study offers a possible tool for monitoring algae blooms and predicting the production of toxin before it occurs."
Harmful algal blooms are difficult to predict, and the bloom-causing organisms typically possess very complex, large genomes. Study authors say the biggest implication will be the ability to look at a bloom on the genetic level. Knowledge of the genes involved in domoic acid production will allow for genetic monitoring of algal blooms and aid in identifying conditions that trigger toxin production.
"Because the genomes of the algae are so complex, the biosynthetic pathways for marine microalgal toxins have remained elusive for quite some time," said senior author Bradley Moore, a chemist and geneticist at Scripps and UC San Diego's Skaggs School of Pharmacy and Pharmaceutical Sciences. "Now that we have both a genome for Pseudo-nitzschia and a genetic pathway for domoic acid production, we are beginning to understand why these microalgae make a toxin and how that capability is activated. This new knowledge may ultimately better educate us how to predict and prepare for future toxic events."
This work advanced research completed in 2011 by David Hutchins of the University of Southern California, also a co-author of this study, that found that when phosphate is limited and the amount of carbon dioxide in the ocean is increased, diatoms can make a large amount of domoic acid and become harmful. The finding was significant in part because scientists have observed that the oceans have been taking on additional amounts of carbon dioxide beyond natural levels due to societal use of fossil fuels. This and rising ocean temperatures mean that domoic acid events are becoming more prevalent, more toxic, and lasting longer than in previous decades. Scripps researchers used the results of Hutchins' study to identify the genes responsible for the production of this toxin.
"We found it very interesting that a combination of phosphate limitation and increased carbon dioxide could have such a strong yet nuanced effect on domoic acid production in culture," said Andrew E. Allen, an ecologist and diatom genomics expert with dual appointment at Scripps and JCVI who is also a senior author on the study. "We were able to directly correlate gene expression to toxin production, and this observation led us straight to the genes encoding domoic acid."

Researchers at JCVI working in Allen's lab extracted and sequenced RNA transcripts from the microalgae, an approach that measures genes that are active. Subsequent analysis of the genetic sequences encoded by the RNA transcripts identified the genes presumed to produce the toxins. In vitro biochemistry experiments conducted in Moore's lab at Scripps then established a series of enzymes that create the core structure of the toxin.
"Some of the domoic acid biosynthetic enzymes that build this toxin are unique at the genetic and biochemical level," said Shaun McKinnie, a postdoctoral researcher at Scripps and the Center for Marine Biotechnology and Biomedicine and co-lead author of the study. "Now that we can link these diagnostic chemical transformations to their enzymes and genes, we're hopeful that researchers can begin to predict domoic acid toxicity potential in a harmful algal bloom to complement current monitoring approaches."
Researchers who study the monitoring and forecasting of harmful algal blooms say this finding offers hope for increased understanding of the phenomenon and can help better project the trajectory of domoic acid events in response to future climate change.
"This breakthrough marks a clear inflection point in our understanding of these events given that so much of the uncertainty surrounding toxic blooms of Pseudo-nitzschia is a result of our incomplete grasp of domoic acid synthesis itself," said biological oceanographer and harmful algal bloom researcher Clarissa Anderson, director of the Southern California Coastal Ocean Observing System (SCCOOS) at Scripps, who was not involved in the study. "The holy grail of harmful algal bloom forecasting on the West Coast and elsewhere is predicting when, where, and ultimately, why Pseudo-nitzschia turns on or turns off domoic acid production. We may never be able to prevent this type of harmful algal bloom, but we can better monitor the early stages of domoic acid production."
The study, "Biosynthesis of the neurotoxin domoic acid in a bloom-forming diatom," was five years in the making.
More information: J.K. Brunson el al., "Biosynthesis of the neurotoxin domoic acid in a bloom-forming diatom," Science (2018). science.sciencemag.org/cgi/doi … 1126/science.aau0382
G. Pohnert el al., "The making of a plankton toxin," Science (2018). science.sciencemag.org/cgi/doi … 1126/science.aau9067
Journal information: Science
Provided by University of California - San Diego

















