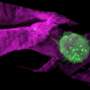
Infrared beams show cell types in a different light

By shining highly focused infrared light on living cells, scientists at the U.S. Department of Energy's Lawrence Berkeley National Laboratory (Berkeley Lab) hope to unmask individual cell identities, and to diagnose whether the cells are diseased or healthy.
They will use their technique to produce detailed, color-based maps of individual cells and collections of cells – in microscopic and eventually nanoscale detail – that will be analyzed using machine-learning techniques to automatically sort out cell characteristics.
Using microscopic color maps to unlock cell identity
Their focus is on developing a rapid way to easily identify cell types, and features within cells, to aid in biological and medical research by providing a way to probe living cells in their native environment without harming the cells or requiring obtrusive cell-labeling techniques.
"This is totally noninvasive," said Cynthia McMurray, a biochemist and senior scientist in Berkeley Lab's Molecular Biophysics and Integrated Bioimaging Division who is leading this new imaging effort with Michael Martin, a physicist and senior staff scientist at Berkeley Lab's Advanced Light Source (ALS).
The ALS has dozens of beamlines that produce beams of intensely focused light, from infrared to X-rays, for a broad range of experiments.
"We're looking at the signatures that define what a cell is. We're interested in cell identity and what determines it," McMurray said. "I've been involved in looking at tissue and the differences in diseased and normal states for a long time, and what I realized is that we could use these infrared beamlines at the ALS to come up with signatures for these cells."
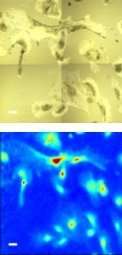
Researchers also plan to use the technique to identify and decipher the molecular properties of microbes and plants.
A goal of the infrared-imaging research, which builds on Berkeley Lab's strengths in neuroscience, infrared imaging, computation, visualization, engineering, and machine learning, is to find out whether a cell's specialization is imposed by its environment or whether it is part of the cell's inherent identity, she said.
The research could help light a path toward controlling the specialization of cells, for example.
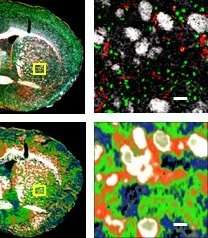
To verify that the technique works, the team is comparing their infrared-based images side-by-side with images captured using a more conventional imaging technique known as immunofluorescence.
Already, through unpublished research the team has found differences in the same types of brain cells taken from different locations in the brain. "On the surface they don't look any different, but if you probe it with infrared light, something about them is different," she said. The imaging technique can be used to show the location and variety of cell types and cell signatures present in tissue samples. "We are very interested in the interactivity of cells."
Initiative targets disease
Long-term goals for the research are to understand disease progression at a cellular level, and to explore how stem cells transform into other cell types. "We want to watch cells differentiate. What allows cells to make the choice to be something else?" she said.
Berkeley Lab scientists received a round of seed money through the philanthropic Chan Zuckerberg Initiative DAF (CZI), an advised fund of Silicon Valley Community Foundation, to support their effort, dubbed "spectral phenotyping."
An Aug. 8 news article in the journal Science highlighted their work and that of a larger research project called the Human Cell Atlas that aims to provide "a unique ID card for each cell type," as well as a 3-D map of how cells form tissues, and new insights into disease.
The Chan Zuckerberg Initiative, launched by Facebook founder Mark Zuckerberg and Priscilla Chan, a pediatrician and philanthropist who is married to Zuckerberg, has three focus areas: science, education, and justice and opportunity. The stated goal of the science focus is to support interdisciplinary teams in developing "science and technology that will help make it possible to cure, prevent, or manage all diseases by the end of the century."
A key to the infrared-imaging technique, McMurray said, is that it allows cells to exist in their native environment, and doesn't require any labels or special prepping that could damage or otherwise alter the cells.
A global focus
Martin, who is part of the infrared-based cell-imaging research, noted that the properties of infrared light produced at the ALS are rare among the world's research institutions, and its possible applications in cell identification are largely unexplored.
The detectors at one of the ALS infrared beamlines are capable of scanning across 1,000 wavelengths for each pixel of the detector – the pixel is the smallest unit of the image – so the cell imaging can generate a large volume of data.
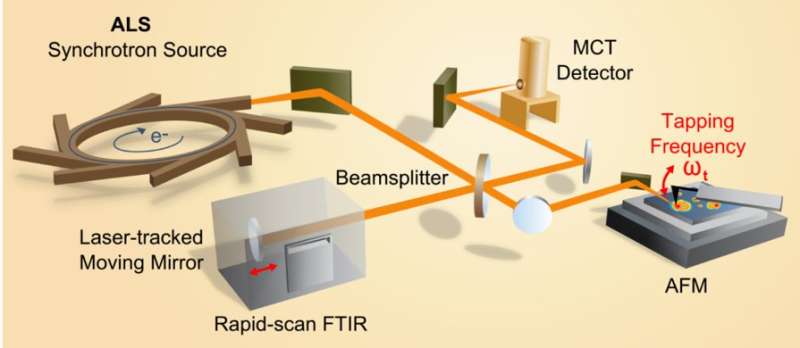
In designing computer algorithms to sort out cells' differences, Martin said, "It's not just about picking one feature. We think it's more powerful to ask broader questions: What's the whole suite of differences?"
By looking across a wide range of infrared wavelengths in each cell image, "Hopefully we'll find something much deeper," he said.
There are plans to use a new, higher-resolution infrared beamline at the ALS that could resolve cell features down to tens of nanometers (billionths of a meter), he also noted, and to develop a way to flow living cells through the infrared beam for high-throughput imaging.
While the properties of infrared light produced at the ALS are relatively unique, Martin and McMurray said it's their hope that the cell-imaging data they generate will prove useful for researchers working with lower-power infrared-imaging tools, too.
The team's proposal to the Chan Zuckerberg Initiative outlines a plan to develop a complete, push-button system that ultimately could be used by the broader research community.
"We want to bring all of this together to develop something that is open to the rest of the world," Martin said.
Provided by Lawrence Berkeley National Laboratory