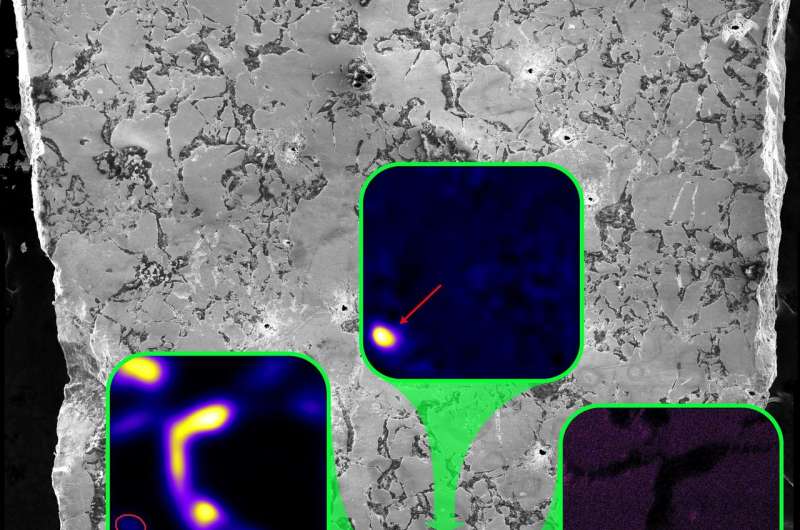
A scanning electron microscope image of uranium ore from the Oklo natural nuclear reactor. Insets show fissionogenic Cs and Ba hotspots. Credit: PNAS
A team of researchers from the U.S. Naval Research Laboratory and Washington University has learned more about possible ways to store modern nuclear waste by studying an ancient natural fission reactor. In their paper published in Proceedings of the National Academy of Sciences, the group describes their study of cores taken from the natural Oklo nuclear reactor and what they found.
As scientists continue to search for new more environmentally friendly ways to produce power, the old ways continue to generate waste. One such source is waste from nuclear power plants. Various options regarding how to store it long term have been discussed, but few have panned out, leaving waste to be temporarily stored onsite. In this new effort, the researchers sought to learn more about what actually happens as nuclear waste decays over its active lifetime. To learn more, they traveled to Gabon, located in West Africa. At a location known as Oklo, there exists the remains of a natural nuclear reactor. Due to a variety of events, the site was the scene of naturally occurring fission approximately 2 billion years ago. The uranium-235 that drove the reactions has long since decayed, but the history of how that occurred remains.
To learn more about what happened as the fissionable material decayed, the researchers took core samples and brought them back to their lab, which houses the Naval Ultra Trace Isotope Laboratory's Universal Spectrometer. There, they were able to piece together the history of the radioactive material as it moved through its elemental states, some of which included isotopes. Of utmost concern was what became of the cesium that was produced as a byproduct of uranium fission. Cesium has been found to be particularly hazardous due to its high degree of radioactivity—it was released into the environment after both the Fukushima and Chernobyl accidents. The researchers found that it was absorbed by an element called ruthenium, approximately five years after the reactor ceased. It was held there in place for almost 2 billion years.
The researchers suggest that discovering that cesium had been contained by ruthenium offers some ideas on possible ways to deal with waste produced in modern reactors. They further note that ruthenium is too rare to use, but something like it might do the trick. They plan to investigate further.
More information: Evan E. Groopman et al. Discovery of fissionogenic Cs and Ba capture five years after Oklo reactor shutdown, Proceedings of the National Academy of Sciences (2018). DOI: 10.1073/pnas.1807267115
Abstract
Understanding the release and sequestration of specific radioactive signatures into the environment is of extreme importance for long-term nuclear waste storage and reactor accident mitigation. Recent accidents at the Fukushima and Chernobyl nuclear reactors released radioactive 137Cs and 134Cs into the environment, the former of which is still live today. We have studied the migration of fission products in the Oklo natural nuclear reactor using an isotope imaging capability, the NAval Ultra-Trace Isotope Laboratory's Universal Spectrometer (NAUTILUS) at the US Naval Research Laboratory. In Oklo reactor zone (RZ) 13, we have identified the most depleted natural U of any known material with a 235U/238U ratio of 0.3655 ± 0.0007% (2σ). This sample contains the most extreme natural burnup in 149Sm, 151Eu, 155Gd, and 157Gd, which demonstrates that it was sourced from the most active Oklo reactor region. We have discovered that fissionogenic Cs and Ba were captured by Ru metal/sulfide aggregates shortly following reactor shutdown. Isochrons from the Ru aggregates place their closure time at 4.98 ± 0.56 y after the end of criticality. Most fissionogenic 135Ba and 137Ba in the Ru migrated and was incorporated as Cs over this period. Excesses in 134Ba in the Ru point to the burnup of 133Cs. Cesium and Ba were retained in the Ru despite local volcanic activity since the reactor shutdown and the high level of activity during reactor operation.
Journal information: Proceedings of the National Academy of Sciences
© 2018 Phys.org