Shining a light on gene regulation
Cancer treatments—from radiation to surgery to chemotherapy—are designed to remove or kill cancerous cells, but healthy cells often become collateral damage in the process. What if you could use lasers to pinpoint the treatment area and deliver medicine to cancer cells only?
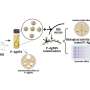
A research team at the University of Delaware, led by Emily Day, an assistant professor of biomedical engineering, is laying the groundwork for a method to inhibit cancer-promoting genes in cancer cells while leaving healthy cells intact. In a new paper published in Nano Letters, the team reveals unprecedented insights into this promising method, which involves coating nanoparticles with gene-regulatory agents and then exposing them to a dose of laser light to unleash that material.
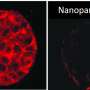
Because the nanoparticles hold the gene regulatory agents inactive until their release is triggered on-demand with light, they have substantial potential to enable high precision cancer therapy while minimizing impact to non-irradiated healthy cells. Day and her team tested their new method against brain tumor cells by using the nanoparticles to silence the expression of green fluorescent protein (GFP) in the cells.
A new way to combine lasers and nanoparticles
Like scalpels and sutures, lasers are often used in medical procedures. Ophthalmologists use lasers to reshape corneas in vision-correcting eye surgery. Dermatologists use lasers to fade scars on skin. Cardiologists use lasers to open clogged arteries. Oncologists use lasers to destroy cancerous cells and tumors.
The Day Lab aims to combine lasers with light-responsive gene regulatory nanoparticles to strategically annihilate cancer cells and minimize collateral damage.
They start with small interfering RNAs, known as siRNAs, which are molecules consisting of complementary strands of RNA that can reduce the expression of certain genes in cancer cells. The siRNAs are coated on nanoparticles made of silica and gold, which measure smaller than the width of a human hair. These particles protect the siRNAs until they reach the desired treatment area. The question is—how do you release them at exactly the right spot?
"Previously, if people wanted to deliver RNA with nanoparticles, they might coat the nanoparticles with a polymer or cell-penetrating peptide, an agent to facilitate cell uptake and delivery," Day said. "The problem is that those agents aren't specific. You end up getting gene regulation in all cells, not just the cancer cells you intended to target for gene regulation."
That's where the precision of lasers comes in.
"Light-triggered siRNA release enables you to silence genes only where the particles and light are combined," Day said. "Since you choose where the light goes, and thus where the siRNA is released, you can achieve very specific gene silencing in tumor cells, reducing the potential for off-target effects."
For these experiments, Day and her team (led by Department of Biomedical Engineering graduate student Rachel Riley) exposed the siRNA-coated nanoparticles to either continuous wave light or short pulses of light administered every one quadrillionth of a second. Both types of irradiation triggered the release of siRNA in double-stranded and single-stranded forms, which came as a surprise since previous studies in the scientific literature suggested that continuous wave irradiation would release only single strands of siRNA (meaning that the two complementary strands of the siRNA separated from each other, rather than the entire duplex releasing from the nanoparticle surface).
"This is the first time anyone has shown that you can get both siRNA duplexes and single strands of RNA to release from nanoparticles using a continuous wave laser as the excitation source," said Day. "However, our studies also reveal that pulsed light is much more efficient at releasing siRNA duplexes than continuous wave light. It is imperative that primarily duplexes be released from the nanoparticles because only duplexed siRNA will function inside cells to silence expression of the targeted genes."
Day continued, "Further, our studies show that pulsed light doesn't heat up cells like continuous wave light does. This is important since too much heat can cause cells to be damaged by mechanisms other than the desired gene regulation."
Her lab group is well-equipped to uncover such insights regarding the mechanism of light-triggered RNA release from nanoparticles because they perform unique interdisciplinary research.

"My lab has a lot of expertise in RNA delivery and cancer biology, so we have a lot more molecular biology skills than a traditional nanotechnology lab might have, and this enables us to study the function of RNA nanocarriers with unprecedented detail," she said.
The new insight that Day's research sheds on the use of continuous wave light versus pulsed light for triggered siRNA release from nanoparticles is an important contribution to the field of drug delivery. In addition to demonstrating the functional differences between the two modes of light excitation, the team also showed that siRNA released from nanoparticles upon exposure to pulsed light reduced the expression of the targeted gene (green fluorescent protein) better than a commercially available material often used to deliver siRNA into cells in lab experiments. This was an exciting observation demonstrating the potential impact of this new system.
This work was conducted in collaboration with Lars Gundlach in the Department of Chemistry and Biochemistry and the Department of Physics and Astronomy at UD, which owns the pulsed laser used in these experiments. Much more research is needed before this could be translated to a viable treatment for cancer patients.
What's next?
"The next step would be to show that we can do this with a more functional and biologically relevant RNA cargo than the siRNA targeting green fluorescent protein that was used in this work," Day said. "There are a lot of different genes that are known to control cancer growth, so we will deliver RNA that targets genes that we know to be important in tumor progression and see if we are delivering enough to have an impact."
The team is particularly interested in studying the delivery of siRNA targeting beta-catenin, which plays an important role in triple-negative breast cancer, and also the delivery of microRNA (which is similar to siRNA, but can regulate multiple genes simultaneously). For example, Day's team hopes to deliver miR-34a, which is considered a master regulator of gene expression in cancer.
In addition to Day and Gundlach, the paper's authors include Rachel Riley, a doctoral student in the Department of Biomedical Engineering; Megan Dang, a doctoral student in Department of Biomedical Engineering; Maggie Billingsley, a senior undergraduate student in the Department of Biomedical Engineering; Baxter Abraham, a doctoral student in the Department of Chemistry and Biochemistry.
Riley, who will soon begin work as a postdoctoral associate at the University of Pennsylvania, started graduate school at UD because she wanted to do science that could make a real impact on the medical field. After losing an aunt to cancer, she was especially motivated to put her talents toward research that could someday help patients.
"The Day Lab's idea of incorporating nanoparticles for specific delivery of therapeutic agents was a really unique approach," she said.
Billingsley has done research in the Day Lab since her first year at UD and wrote three peer-reviewed papers based on her work in the group. In 2017, she was the first author on a paper published in PLOS One. Next, Billingsley will attend graduate school at the University of Pennsylvania, where she will study immunotherapy. (She and Riley, a duo Day calls a "dream team," could likely work together in the future.)
"When interviewing with graduate schools, this experience with The Day Lab was the main thing that helped me stand out in interviews and also understand what I wanted out of graduate school," Billingsley said. "I have had so many opportunities to go down my own path and also work with others on new methods, ideas, and areas of research."
This paper also gave Billingsley a taste of an important aspect of scientific discovery.
"We found some results that conflicted with literature, which was a new experience for me," she said. "That was more exciting that just confirming what was expected."
Overall, Day and her team are excited to further pursue this technology to reveal its potential as a high precision cancer therapy.
More information: Rachel S. Riley et al. Evaluating the Mechanisms of Light-Triggered siRNA Release from Nanoshells for Temporal Control Over Gene Regulation, Nano Letters (2018). DOI: 10.1021/acs.nanolett.8b00681
Journal information: Nano Letters , PLoS ONE
Provided by University of Delaware