Bacterial toxins wreak havoc by crippling cellular infrastructure
Bacterial toxins can wreak mass havoc within cells by shutting down multiple essential functions at once, a new study has found.
The discovery could one day open the door to exploring better ways to fight life-threatening infections, said lead researchers Elena Kudryashova and Dmitri Kudryashov of The Ohio State University.
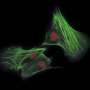
The study focused on how one bacterial toxin called ACD (a type linked to cholera and poisoning by raw oysters) modifies an abundant cellular protein called actin and converts it into a secondary toxin. Actin is involved in a host of processes including muscle contraction, cell division, cellular communication and immune response.
The researchers wanted to better understand how a relatively small amount of bacterial toxin could do such swift, significant damage to a strong network of actin. Their study appears in the journal Current Biology.
In theory, the actin's abundance within every single human cell should make it a difficult to disable - and disabling targets is the business of a bacterial toxin looking to gum up the immune system and make a human or animal sick.
"You can think of actin like the bones and the muscles of the cell - and a barrier to what comes into and out of the cell," said Kudryashov, an associate professor of chemistry and biochemistry at Ohio State. "It's clearly beneficial for bacteria to paralyze it in some way and we now think we know how that happens for one actin-specific toxin."
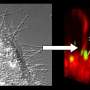
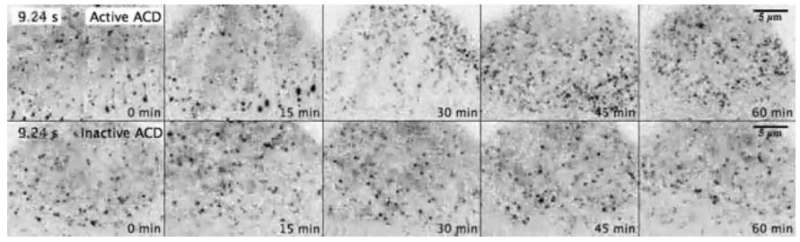
When the Ohio State research team looked at what was happening in real time in a living cell, they were able to understand how quickly and efficiently the toxin took over - and how that happened.
The toxin cripples the action of "instructor" proteins, many of which share a common property - the ability to bind several actin molecules at once. By permanently linking together several actin molecules, ACD toxin converts them into a new universal toxin that binds tightly to those "instructors" and blocks their activity.
This amplifies toxicity and disorganizes the cell, and it doesn't take much. A tiny fraction of actin - about 2 percent within a given cell - is affected. The toxin is then redirected toward less-abundant targets, which leads to a cascade of cellular changes that break down normal function. Previous work led to a better understanding of this activity outside of a living cell.
"This is basically like crippling a nation by disabling all its instructors: political and military leadership, teachers and others. The population (actin) is there, but without proper instructions most transportation, imports and exports, border protection and other normal activities are halted or disorganized all at once," Kudryashov said.
Work such as this is especially important in light of growing concerns about antibiotic-resistant infections, said Kudryashova, a research scientist in chemistry and biochemistry at Ohio State.
"The bacteria are becoming 'smarter,' and so we must become much smarter as well," she said.
Toxins are the key to bacteria's power to make people and animals sick. A single molecule of some of the most lethal toxins - those released by bacteria that cause whooping cough and dysentery, for example - can kill an entire cell.
"When antibiotics worked effectively, we worried less about these processes in the cell, but today we must better understand how bacteria thrive in our bodies so that scientists might someday find alternative ways to fight them," Kudryashova said.
The research team's previous work found that this particular bacterial toxin goes after the abundant actin proteins as opposed to the more-expected targeting of less-common molecules that send important cellular signals.
More information: Actin Cross-Linking Toxin Is a Universal Inhibitor of Tandem-Organized and Oligomeric G-Actin Binding Proteins, Current Biology (2018). DOI: 10.1016/j.cub.2018.03.065
Journal information: Current Biology
Provided by The Ohio State University