A model for autoignition in turbulent jets
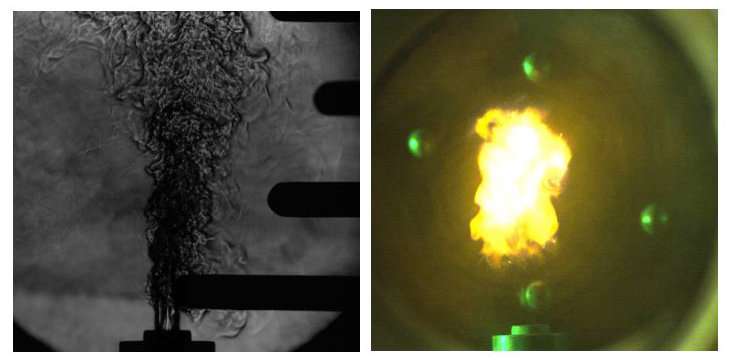
Jets are rapid streams of liquids or gases that forcefully shoot into a surrounding medium. When ignitable substances are involved, combustion—rapid chemical reactions that result in heat and light—can occur. Combustion in jets has many industrial and technological applications, and is thus of great interest to scientists and engineers.
The chemical interactions in jets with an oxidizing component and a chemically-reacting agent can either produce a weak reaction inducing slow oxidation in the reactive component, or develop quickly and instigate thermal runaway, which results in a swift temperature increase that spontaneously triggers combustion. Autoignition ensues when this spontaneous combustion results in a visible flame. In a paper published earlier this week in the SIAM Journal on Applied Mathematics, Peter V. Gordon, Uday G. Hegde, and Michael C. Hicks present a mathematical model for autoignition in free round turbulent jets.
The mathematics of autoignition in reactive materials dates back to the 1920s and 30s, particularly to the early work of Nikolay Semenov, David Frank-Kamenetskii, and Yakov Borisovich Zel'dovich. Their research established a mathematical combustion theory called the theory of thermal explosion, and subsequent studies were typically based on their findings. One common truth characteristically unites all thermal explosion studies: prior to autoignition, the reactive systems' dynamics are fairly straightforward. As a result, scientists can simplify a system of equations governing the evolution of reactive systems to create and examine models of autoignition in great detail.
Gordon et al. use recent experimental advances in the study of hydrothermal flames to analyze autoignition in free jets. First observed about 30 years ago, hydrothermal flames arise in aqueous (water) environments at conditions above the thermodynamical critical point of water. They are a key component of an emerging "green" water purification technology called supercritical water oxidation (SCWO), and occur spontaneously during SCWO via autoignition. "The principal advantage of this technology is that it allows almost perfect conversion rates of organically-contaminated waste streams without producing harmful intermediate species," Hicks said. "The presence of hydrothermal flames in SCWO devices is often desirable since it allows for substantially-reduced reaction times—from seconds to milliseconds—thereby dramatically enhancing decomposition rates."
Experimental studies of hydrothermal flames typically involve a closed combustion vessel with an injection inlet. The authors derive an elementary autoignition model for a fully-developed, round turbulent reactive jet. The jet is formed via injection of fuel and oxidizer into the vessel, which contains pure water at a supercritical state at rest. The injected stream creates a round jet that is either laminar (smooth with parallel flow) or turbulent (irregular). When conditions are right, the jet autoignites axially downstream from the injection point.
To effectively illustrate autoignition, Gordon et al. make certain assumptions about the jet's form and overall conditions. "The key experimental facts that we use in our theory is that the shape of the jet, as well as the velocity and concentration fields of the species within the jet prior to autoignition, can be viewed as a priori prescribed," Gordon said. "Specifically, in a first approximation, the main region of the jet assumes the shape of a conical frustum (a cone with the pointed top sliced off). Moreover, the velocity within the main part of the jet—in the direction perpendicular to the jet—is negligible in comparison to one in the direction of injection. The latter is radially symmetric and inversely proportional to the distance from the injection point, and the same applies to concentration fields of reactive and oxidizing components within the jet."
Using experimental observations and the aforementioned assumptions, the authors separate the model's hydrodynamic and reactive components. This drastically simplifies autoignition, reducing it to one differential equation. "The problem reduces to the analysis of a single equation that describes evolution of the temperature field within the jet, which we can analyze using a general framework of the Frank-Kamenetskii theory of thermal explosion," Gordon said. "This leads to sharp characterization of an autoignition event in terms of principal physicochemical and geometric parameters."
Gordon et al.'s model is a counterpart to their previous model of autoignition for laminar co-flow jets, and reveals some valuable truths about autoignition. "The results of the model's analysis allow us to correlate specific values of principal physicochemical and geometric parameters of the problem with the event of autoignition, or absence thereof," Hegde said. "This, in turn, allows one to identify parametric regimes where autoignition takes place, and therefore can be used in guiding experimental studies of hydrothermal flames."
The authors' conclusions will serve the experimental studies of scientists exploring the relation between hydrothermal flames and autoignition. "This work is applicable in the design of next-generation SCWO reactors that will rely on the spontaneous ignition and subsequent control of hydrothermal flames to sustain temperatures and reaction kinetics of SCWO processes in real world applications, like waste remediation and water reclamation," Hicks said. Such research is transpiring at NASA's Glenn Research Center, in Cleveland, Ohio.
"We are currently conducting laboratory experiments with hydrothermal flames in organically-contaminated environments to verify the model predictions," Hegde said. "Qualitatively, we have already seen good agreement with predicted model trends. Quantitative comparisons are more challenging due to the technical difficulties of making accurate in situ measurements in SCWO environments, and are the subject of ongoing and future work."
More information: Peter V. Gordon et al. An Elementary Model for Autoignition of Free Round Turbulent Jets, SIAM Journal on Applied Mathematics (2018). DOI: 10.1137/17M1147780
Journal information: SIAM Journal on Applied Mathematics
Provided by Society for Industrial and Applied Mathematics