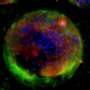
Artificial and biological cells work together as mini chemical factories

Researchers have fused living and non-living cells for the first time in a way that allows them to work together, paving the way for new applications.
The system, created by a team from Imperial College London, encapsulates biological cells within an artificial cell. Using this, researchers can harness the natural ability of biological cells to process chemicals while protecting them from the environment.
This system could lead to applications such as cellular 'batteries' powered by photosynthesis, synthesis of drugs inside the body, and biological sensors that can withstand harsh conditions.
Previous artificial cell design has involved taking parts of biological cell 'machinery' - such as enzymes that support chemical reactions - and putting them into artificial casings. The new study, published today in Scientific Reports, goes one step further and encapsulates entire cells in artificial casings.
The artificial cells also contain enzymes that work in concert with the biological cell to produce new chemicals. In the proof-of-concept experiment, the artificial cell systems produced a fluorescent chemical that allowed the researchers to confirm all was working as expected.
Lead researcher Professor Oscar Ces, from the Department of Chemistry at Imperial, said: "Biological cells can perform extremely complex functions, but can be difficult to control when trying to harness one aspect. Artificial cells can be programmed more easily but we cannot yet build in much complexity.
"Our new system bridges the gap between these two approaches by fusing whole biological cells with artificial ones, so that the machinery of both works in concert to produce what we need. This is a paradigm shift in thinking about the way we design artificial cells, which will help accelerate research on applications in healthcare and beyond."
To create the system, the team used microfluidics: directing liquids through small channels. Using water and oil, which do not mix, they were able to make droplets of a defined size that contained the biological cells and enzymes. They then applied an artificial coating to the droplets to provide protection, creating an artificial cell environment.
They tested these artificial cells in a solution high in copper, which is usually highly toxic to biological cells. The team were still able to detect fluorescent chemicals in the majority of the artificial cells, meaning the biological cells were still alive and functioning inside. This ability would be useful in the human body, where the artificial cell casing would protect the foreign biological cells from attack by the body's immune system.
First author of the study Dr Yuval Elani, an EPSRC Research Fellow also from the Department of Chemistry, said: "The system we designed is controllable and customisable. You can create different sizes of artificial cells in a reproducible manner, and there is the potential to add in all kinds of cell machinery, such as chloroplasts for performing photosynthesis or engineered microbes that act as sensors."
To improve the functionality of these artificial cell systems, the next step is to engineer the artificial coating to act more like a biological membrane, but with special functions.
For example, if the membrane could be designed to open and release the chemicals produced within only in response to certain signals, they could be used to deliver drugs to specific areas of the body. This would be useful for example in cancer treatment to release targeted drugs only at the site of a tumour, reducing side effects.
While a system like that may be a way off yet, the team say this is a promising leap in the right direction. The work is the first example of fusing living and non-living components to emerge from Imperial and King's College's new FABRICELL centre for artificial cell science.
More information: Yuval Elani et al, Constructing vesicle-based artificial cells with embedded living cells as organelle-like modules, Scientific Reports (2018). DOI: 10.1038/s41598-018-22263-3
Journal information: Scientific Reports
Provided by Imperial College London