A milk protein could be used to give foods cancer-fighting, immune-boosting properties
Scientists from A*STAR have developed a system to transport a functional protein to the tissues of the digestive tract where it may confer a range of health benefits. This overcomes previous obstacles where the molecules broke down before they could reach their target receptors.
Lactoferrin, found naturally in breastmilk, is a biologically active protein that provides vital support during early infancy. It also has antimicrobial, anti-inflammatory and cancer-fighting properties, and can even help with weight loss. Adults, however, cannot benefit from dietary lactoferrin because the digestive enzymes and acid in their mature stomachs degrade the protein before it can reach the small intestine. It's there, says A*STAR's Maxim Kiryukhin, that lactoferrin receptors are thought to absorb the protein and transport it to the lymphatic system, where it provides its beneficial effects.
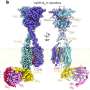
Kiryukhin, from the Institute of Materials Research and Engineering, and his team have succeeded in protecting lactoferrin molecules on their journey through the stomach by coating them in layers of gastric-resistant casing. "We start with calcium carbonate microparticles. These are porous, and absorb lactoferrin to produce a 'core' around which we can build our protective shell," says Kiryukhin. "Then, we dissolve the calcium carbonate, leaving the lactoferrin contained within micrometer-sized shells, designed to be stable within gastric fluid, but to degrade in intestinal fluid."
The shell utilizes eight alternating layers of bovine serum albumin and tannic acid grouped into four 'bilayers," which, aside from offering protection, are also safe and cheap. Tannic acid also has its own antioxidant properties.
Kiryukhin's team then performed both in vitro and in vivo studies, first using simulated digestive enzymes to verify the capsules' desired properties, before testing them in mice. In their mouse model, the scientists fluorescently labeled the protein so they could track it through the stages of digestion. They found high levels of lactoferrin in the small intestine and liver, verifying that the protein survived gastric digestion and was absorbed by the tissues of the intestine.
Kiryukhin says that his team is now assessing whether their process is commercially viable: "We're in discussions with several companies with regard to how we can move our technology forward."
More information: Ece Kilic et al. Formulation for Oral Delivery of Lactoferrin Based on Bovine Serum Albumin and Tannic Acid Multilayer Microcapsules, Scientific Reports (2017). DOI: 10.1038/srep44159
Journal information: Scientific Reports