Saliva proteins could explain why some people overuse salt
Many Americans consume too much salt. Now in a study appearing in the Journal of Agricultural and Food Chemistry, scientists report that people who can easily taste salt have differing amounts of certain proteins in their saliva than those who are less sensitive. The finding could help explain why some of us have a hard time shaking the salt habit and could potentially lead to the development of more desirable low-sodium foods.
Consuming too much sodium increases the risk of heart disease and stroke, two of the leading causes of death in the U.S. Some lower sodium products are on the market, but many consumers avoid these foods because they think the foods won't be tasty enough or flavorful enough. To develop more palatable products, researchers are trying to gain a better understanding of how the body processes and perceives saltiness. Although saliva is thought to play a role, it's unclear exactly what components of the liquid could explain the differences in salt perception among people. In a small study, Thomas Hofmann and colleagues sought to fill that knowledge gap.
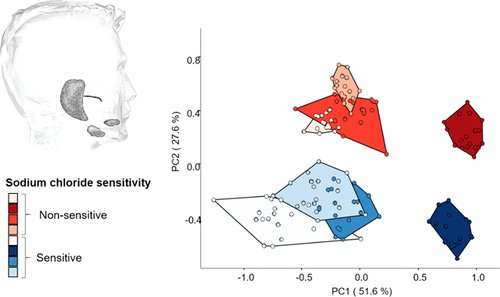
The researchers classified volunteers into sensitive and non-sensitive groups according to how salty the participants thought various sodium chloride solutions were. Using liquid chromatography and mass spectrometry, the team identified several salivary proteins that differed between those who could readily detect salt and those who couldn't. Surprisingly, they found the largest differences in the resting saliva of the subjects compared to saliva produced after swishing around a salty solution. In their resting saliva, sensitive subjects had higher amounts of endopeptidases, enzymes that cut up proteins, than non-sensitive subjects. The researchers suggest that the enzymes could be modifying sodium channels, which would increase the amount of sodium that gets into cells. Alternatively, the enzymes could be cutting proteins in the saliva to produce salt-enhancing peptides in people who are sensitive.
More information: Theresa Stolle et al. Salivary Proteome Patterns Affecting Human Salt Taste Sensitivity, Journal of Agricultural and Food Chemistry (2017). DOI: 10.1021/acs.jafc.7b03862
Abstract
To investigate the role of perireceptor events in inter-individual variability in salt taste sensitivity, 31 volunteers were monitored in their detection functions for sodium chloride (NaCl) and classified into sensitive (0.6–1.7 mmol/L), medium-sensitive (1.8–6.9 mmol/L), and nonsensitive (7.0–11.2 mmol/L) subjects. Chemosensory intervention of NaCl-sensitive (S+) and nonsensitive (S–) panellists with potassium chloride, ammonium chloride, and sodium gluconate showed the salt taste sensitivity to be specific for NaCl. As no significant differences were found between S+ and S– subjects in salivary sodium and protein content, salivary proteome differences and their stimulus-induced dynamic changes were analyzed by tryptic digestion, iTRAQ labeling, and liquid chromatography–tandem mass spectrometry analysis. Differences in the salivary proteome between S+ and S– subjects were found primarily in resting saliva and were largely independent of the dynamic alterations observed upon salt stimulation. Gene ontology enrichment analysis of key proteins, i.e., immunoglobulin heavy constant y1, myeloblastin, cathepsin G, and kallikrein, revealed significantly increased serine-type endopeptidase activity for the S+ group, while the S– group exhibited augmented cysteine-type endopeptidase inhibitor activity by increased abundances in lipocalin-1 and cystatin-D, -S, and -SN, respectively. As proteases have been suggested to facilitate transepithelial sodium transport by cleaving the y-subunit of the epithelial sodium channel (ENaC) and protease inhibitors have been shown to reduce ENaC-mediated sodium transport, the differentially modulated proteolytic activity patterns observed in vivo for S+ and S– subjects show evidence of them playing a crucial role in affecting human NaCl sensitivity.
Journal information: Journal of Agricultural and Food Chemistry
Provided by American Chemical Society




















