Credit: Wageningen UR
A bacterium is fulfilling a long-cherished wish of many chemists. The E. coli bacterium, which has been modified to equip it with special enzymes, has been shown to make building blocks for plastics, such as polyesters, directly from petroleum components called alkanes, using very little energy. This study was carried out by Youri van Nuland and he obtained his doctorate for this research at Wageningen University & Research on 20 October.
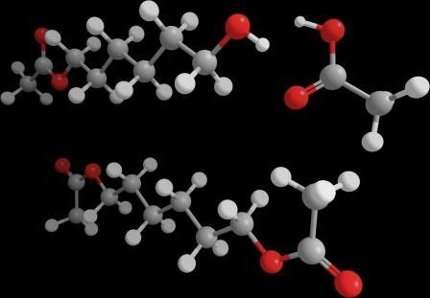
To produce plastics, the building blocks in question must be coupled to one another, each building block needing two special chemical groups, which can be compared to 'hooks' or 'eyes', at its ends. The alkanediols are particularly important building blocks. They are alkanes, such as propane and butane, with two alcohol groups at their ends and they can be used for the production of polyesters, polyurethanes, polyamides and other plastics.
The alkanediol building blocks currently on the market are often made from the more complex raw materials acetylene or benzene, via a number of energy-intensive steps that release large quantities of the greenhouse gases carbon dioxide and laughing gas (nitrous oxide). Six kilograms of CO2 are emitted per kilogram of product. Around 1.8 million tonnes of butanediol are produced annually.
Chemical wish list
An obvious solution would therefore be to make these alkanediol building blocks directly from simple, readily-available alkanes, by equipping both ends of these molecules with an alcohol group or 'hook'. This conversion consumes little energy and releases only limited quantities of greenhouse gases. A conversion method of this kind has therefore been high on the wish list since the birth of the petrochemical industry.
So far, however, the many industrial and university laboratories have been unable to realize a direct route for synthesizing these building blocks for the manufacture of plastics. The problem was that, during the process, the inner carbon atoms of the alkanes were equipped with an alcohol group as well as the outer carbon atoms. This meant that the chemical reaction was not specific enough and yielded undesirable by-products. The alcohol groups were, moreover, converted into acid groups or the whole molecule was burned to carbon dioxide and water.
The enzyme alkane hydroxylase (AlkB) is specifically able to equip only the outer carbon atoms of alkanes with alcohol groups although it also changes the alcohol groups into acid groups. Furthermore, it is only able to do so on one side of the alkane molecule. It looked as though the chemist's wishes were not to be fulfilled; there was a stalemate.
However, Youri van Nuland, a PhD student in the Bioprocess Engineering research group at Wageningen University & Research, has now succeeded in realizing the desired conversion from alkanes to alkanediols using the AlkB enzyme. He genetically modified a strain of the E. coli bacterium and equipped it with AlkB and another enzyme, alcohol acetyltransferase. The Atf1 enzyme rapidly safeguards the alcohol group formed by AlkB, by enabling it to react with acetic acid to form a stable ester before it can be converted further into an acid group. A surprising point here is that AlkB is now able to equip the other end of the alkane with an alcohol group as well and that Atf1 also enables this to react to form an ester. The esters formed from an alkanediol are easy to convert into the required alkanediol and this conversion requires little energy.
Youri van Nuland has demonstrated this conversion on a laboratory scale with alkanes varying from butane to decane. Further optimization and upscaling will be necessary to make the method into an industrial process. A patent is pending for his discovery.
Provided by Wageningen University