Halogen bonding-mediated metal-free controlled cationic polymerization
Chemists at the Nagoya Institute of Technology (NITech) report a metal-free method to control cationic polymerization that provides a new framework for higher quality industrial polymers applicable to semi-conductors and bio-materials. The reaction depends on weak halogen bonding and the addition of a small amount of ammonium salt to produce long, homogeneous polymers. The study can be read in Chemistry - A European Journal.
The polymer is synthesized by cationic polymerization, as an example, through a reaction process that along with the vinyl molecule requires the combination of a catalyst and an initiator called an initiating system.
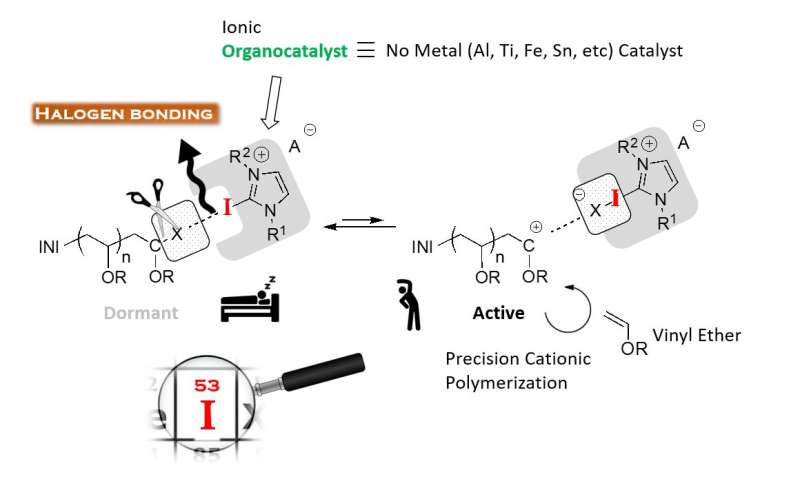
"Metal halide catalysts are well studied in cationic polymerization, but they create impurities responsible for the coloration and degradation of polymers. There is high demand for metal-free initiating systems," says Associate Professor Koji Takagi, who first-authored the study.
The challenge in cationic polymerization is to produce a long polymer that is homogeneous without adding metal-based catalysts. The role of the catalysts is to abstract an anion from the initiator, such as a halide, that reversibly interacts with the catalyst and the polymer propagating end. For this reason, weak bonding is preferred, as otherwise side effects can occur that compromise the homogeneity and size of produced polymers. In the new study, Takagi and his colleagues show that chloride makes the best halide, as it forms appropriately weak halogen bonds with a 2-iodoimidazolium catalyst.
"The 2-iodoimidazolium component is a requirement for the catalyst if we want to polymerize isobutyl vinyl ether (IBVE)," he said, speaking about the first vinyl compound scientists used to demonstrate cationic polymerization.
The researchers found that if other halides were used in the initiator structure, then some of the catalyst would degrade, in part because of the strength of the halogen bond.
The chloride anion used in his initiator could shift back and forth between the catalyst and the propagating end of IBVE efficiently, thus generating a polymer with large molecular weight and narrow molecular weight distribution.
The study further shows the ideal temperature (-10 oC) and concentrations of initiating system for the reaction (10 mM each). These conditions minimized side effects that risked compromising the molecular weight and molecular weight distribution.
Finally, the controllability of cationic polymerization could be enhanced with the addition of a small concentration of ammonium salt.
"Adding ammonium salt allowed us to modify the polymerization rate and molecular weight distribution," said Takagi.
Overall, he remarks that the process has a large number of attractive features for polymer synthesis, and expand the application of the polymer for electrical and bio-material usages.
More information: Koji Takagi et al, Halogen-Bonding-Mediated and Controlled Cationic Polymerization of Isobutyl Vinyl Ether: Expanding the Catalytic Scope of 2-Iodoimidazolium Salts, Chemistry - A European Journal (2017). DOI: 10.1002/chem.201702455
Journal information: Chemistry – A European Journal
Provided by Nagoya Institute of Technology