Chemists explained the origin of the green fluorescence
Researchers at the Lomonosov Moscow State University in cooperation with Danish molecular physicists have revealed the mechanism that determines the sensitivity of green fluorescent protein to light exposure. The scientists have proved that an isolated chromophore group is capable of emitting light outside the protein environment, while the protein function is to enhance its fluorescent properties. The results are published in the Journal of the American Chemical Society.
Proteins from the GFP family are widely used as biomarkers. With their help, researchers could visualize biological processes taking place inside living cells. These proteins are widely used in molecular and cellular biology due to their unique ability to fluoresce upon absorption of light of a certain wavelength.
Chromophore groups, or chromophores, are responsible for light absorption. The chromophore is composed of just three amino acid residues. Until now, researchers believed the protein environment caused its chromophore to emit light under irradiation. This idea was supported by the fact that the isolated chromophore loses its fluorescent properties upon protein denaturation, i.e. unfolding of the protein. For the first time, the authors have predicted and experimentally proved that the isolated chromophore can also fluoresce outside the protein environment, but only at certain temperatures.
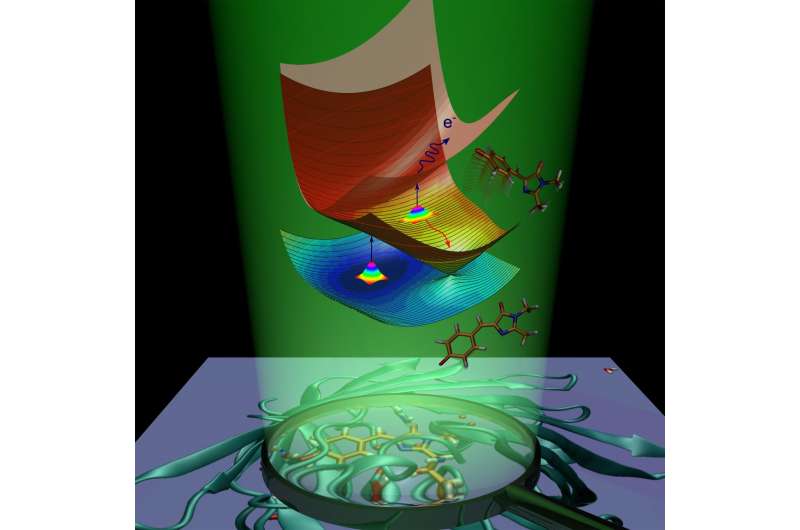
Following absorption of light, the fluorescent protein gains extra energy and undergoes transition to the electronically excited state. The protein molecule is striving to return to the ground state by getting rid of the excess energy. This process is called deactivation and proceeds in two ways. The first one is fluorescence. In transit from the excited state to the ground state, the chromophore emits energy in the form of light, and as a result, the protein becomes luminous.
The second way is called nonradiative relaxation. During this process, electronic excitation energy, transferred to a molecule upon light absorption, is transformed into vibrational energy of nuclei. Internal conversion inside the green fluorescent protein is followed by a chemical reaction—cis-trans isomerization of the chromophore (i.e. intramolecular rotation about one of the double bonds inside the molecule).
In the case of the anionic (negatively charged) chromophore of the green fluorescent protein, energy can also be transferred to one electron, which exits the chromophore. The chromophore, which undergoes isomerization or loses an electron, removes excess electronic energy and doesn't fluoresce. Fluorescence is called a radiative channel of deactivation, while internal conversion and electron emission are called nonradiative channels. The scientists have experimentally used both nonradiative channels to study the excited-state decay and the ground-state recovery of the isolated GFP chromophore at various temperatures.
The scientists have shown that the isolated GFP chromophore can be trapped in the excited state due to the presence of intrinsic energy barriers along the nonradiative deactivation pathways. When the chromophore is cooled to 100 K, the trapping lasts for nanoseconds, thus enabling the slow radiative channel. The trapping barriers prevent the chromophore from returning to the ground state by fast nonradiative conversion and causing the molecule to fluoresce.
Chemist Anastasia Bochenkova of the Lomonosov Moscow State University says, "The GFP chromophore is intrinsically fluorescent. Interactions between the chromophore group and the nearest protein environment improve conditions for trapping the chromophore in the excited state, thus enhancing the molecular fluorescence. As a result, the radiative channel becomes predominant inside the protein already at room temperature."
The scientists have studied the chromophore properties both theoretically and experimentally. The scientists elaborated a theoretical model and estimated an excited-state lifetime of the isolated chromophore, based on the high-level quantum-chemistry calculations. Such calculations require high-performance computing resources, and the scientists used the Lomonosov Supercomputing Center for their studies.
The Danish scientists designed and created a new experimental technique for studying light-induced molecular dynamics of isolated charged chromophores in vacuo—femtosecond pump-probe action spectroscopy with 2-D temporal resolution. Both delay time between two femtosecond laser pulses and decay time of a particular molecular action were varied. The new technique resolves the light-induced dynamics of the GFP chromophore as it happens in different decay channels through both the excited and ground electronic states, depending on temperature. Based on the results, the researchers have concluded that native GFP chromophores are intrinsically able to fluoresce outside the protein environment with a nanosecond lifetime, fully supporting theoretical predictions.
Anastasia Bochenkova says, "Our research aims at disclosing molecular mechanisms of functioning of living systems. A fundamental challenge is to understand the relationship between the properties of an entire light-sensitive biosystem and its much smaller active chromophore, which is at the heart of the molecular machinery. Here, we have shown that the properties of a single chromophore are important for understanding the mechanisms of action of the entire GFP protein. This knowledge is at the intersection of physics, chemistry and biology, and results in revising the prevailing paradigm about the role of the protein environment in the fluorescence of the GFP protein. Beyond solving the question about the origin of the intrinsic fluorescence of GFP, the work shows that detailed molecular knowledge obtained through high-level quantum chemistry calculations may be used together with molecular engineering in designing new functional biomaterials with optimal photoresponse."
More information: Annette Svendsen et al, Origin of the Intrinsic Fluorescence of the Green Fluorescent Protein, Journal of the American Chemical Society (2017). DOI: 10.1021/jacs.7b04987
Journal information: Journal of the American Chemical Society
Provided by Lomonosov Moscow State University