Chromosome mechanics guide nuclear assembly
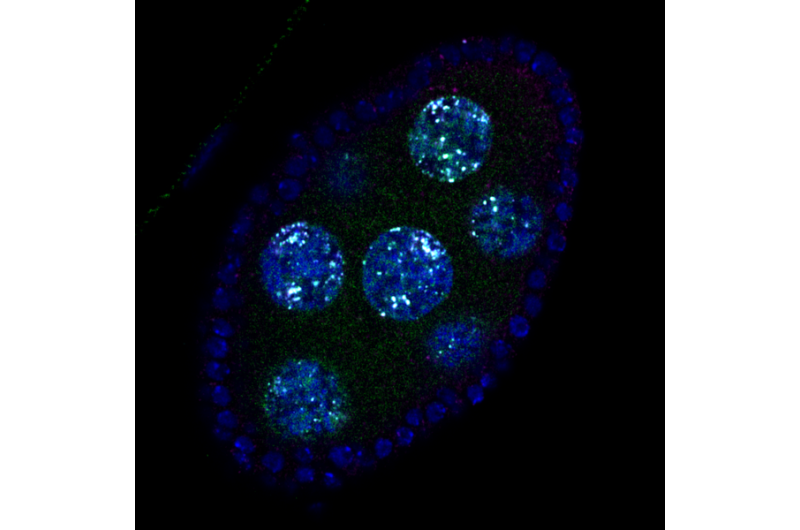
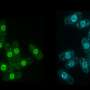
Every one of our cells stores its genome within the nucleus – the quintessential subcellular structure that distinguishes eukaryotic cells from bacteria. When animal cells divide, they disassemble their nucleus, releasing individual chromosomes for proper segregation to daughter cells. At the end of cell division, the daughter cells reassemble a single nucleus around a complete set of chromosomes. The formation of a single nucleus is critical for the maintenance of genomic integrity. Individual chromosomes packaged into separate, small nuclei are prone to massive DNA damage, leading to mutations as well as chromosome rearrangement and loss. Cancer cells often contain small, multiple nuclei, which may drive genome disruption as well as disease progression. Nonetheless, how cells package their genome into just one nucleus has been a mystery.
A genetic screen for micronuclei
The Gerlich lab at the IMBA set out to solve this problem by screening for genes that are required to assemble a single nucleus in human cells. They uncovered "barrier-to-autointegration factor" (BAF), a multifunctional protein that binds DNA as well as many proteins. Without BAF, cells formed fragmented nuclei at the end of cell division. BAF was already known to link DNA to specific proteins at the nuclear membrane. Unexpectedly, Gerlich's lab discovered that nuclear assembly did not require BAF's association with nuclear membrane proteins. Instead, they found that BAF's ability to bind and bridge distant DNA sites was essential to shape a single nucleus.
A dense BAF-DNA network shapes a single nucleus
How does BAF's association with DNA regulate formation of the nucleus? The authors found that BAF forms a compact and mechanically stiff network with DNA. This created a coherent surface mesh around the set of chromosomes, which is impenetrable for nuclear membranes. This barrier prevents membranes from separately enwrapping individual chromosomes, and therefore guides the formation of a single nucleus.
The work, published in the current issue of Cell, answers a fundamental question in biology and reveals an entirely unanticipated function for BAF. "Our findings suggest an entirely new role for DNA cross-bridging beyond genomic functions such as regulating gene expression and recombination, by forming boundaries and mechanical scaffolds of subcellular compartments. We are excited to further uncover the molecular mechanisms that shape mitotic chromosomes and control their interactions with other cellular components" says Gerlich.
-

This drawing illustrates dynamic remodeling in ESCRT-III polymers. Vps4 mediates turnover of ESCRT-III subunits within growing and constricting polymers—analogous to Lego figures exchanging the building blocks within a large spiral assembly. Credit: Beata Edyta Mierzwa, BeataScienceArt.com -
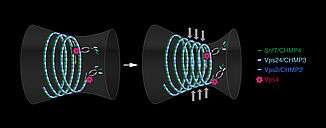
VPS4 mediates turnover of ESCRT subunits with growing and constricting polymers. Credit: IMBA -
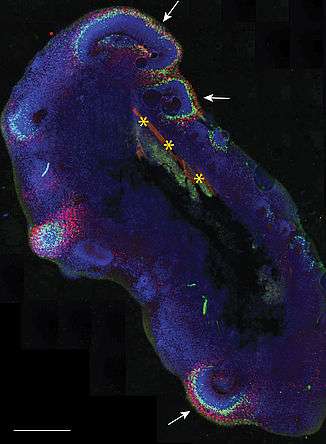
Bioengineered organoids or so called enCORs are supported by a floating scaffold of PLGA-fiber microfilaments. Credit: Institute of Molecular Biotechnology
More information: Matthias Samwer et al. DNA Cross-Bridging Shapes a Single Nucleus from a Set of Mitotic Chromosomes, Cell (2017). DOI: 10.1016/j.cell.2017.07.038
Journal information: Cell
Provided by Institute of Molecular Biotechnology